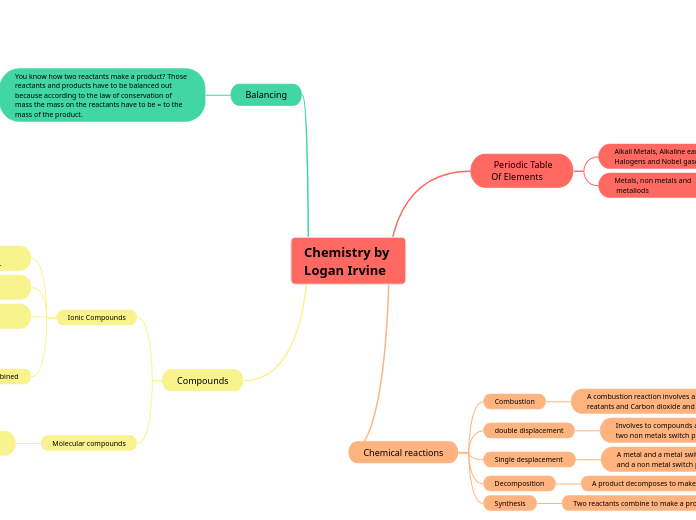
Chemistry by
Logan Irvine
Periodic Table
Of Elements
Alkali Metals, Alkaline earth metals,
Halogens and Nobel gases
Metals, non metals and
metaliods
Metals to the left. metalloids in the centre and non metals on the right.
Chemical reactions
Combustion
A combustion reaction involves a fuel and oxygen as reatants and Carbon dioxide and water as products
Example: CH + O2 is CO2 +HO2 + thermal energy
double displacement
Involves to compounds as reactants. Two metals or two non metals switch places
Example: BaSO + CaNO would be BaNo + CaSO
Single desplacement
A metal and a metal switch places or a non metal and a non metal switch places .
Example: Nal + CaCl will be NalCl + Ca
Decomposition
A product decomposes to make two reactants
Example: H2O is H2 + O2
Synthesis
Two reactants combine to make a product
Example: H2 + O2 is H2O
Balancing
You know how two reactants make a product? Those reactants and products have to be balanced out because according to the law of conservation of mass the mass on the reactants have to be = to the mass of the product.
Example: C+ H2 = C2H8 is not correct because the mass of the reactants does not equal the mass of the product. The correct equation is 3 C 4 H2 = 1 C3H8. In this equation 3 carbons are needed to make a singular C3 and 4 Hydrogen gas is needed to make H8.
Compounds
Ionic Compounds
Valence electron: Number of electrons on outer most layer of diagram. Is also the group number.
Anion is a negatively charged ion and it gets electrons
Cation is a possitivly charged ion and it losses electrons
Metal and non metals combined
Multivalent: use roman numerals to know which one you are using
Example : Fe2O3 is Iron(III) oxide
Polyatomic ion: made up of more than one atom and acts as a single particle
Example: Nitrate is NO3
Rule: Name metal as is
and change the ending of the non metal to ide
Example: CO2 is Carbon dioxide
Molecular compounds
Metal and metal combine of a non metal and non metal combine
Name them by using prefixes and group number
Example: H2O is hydrogen dioxide. if the first element has one valent electron, you don't use mono and just name it as is.