Physiology
The Study of the normal functioning of a living organism and it's component parts including all its chemical and physical processes.
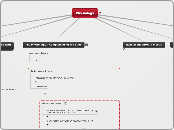
Organ Systems of the Body and their Function
Function and processes are two related concept in physiology:
Function of a physiological event or system refers to why it exists or happens. This is know. as the teleological approach to physiology.
An example of the teleological answer to the question why the RBCs transport oxygen is "because cells need oxygen and the RBCs bring it to the,"
Physiological processes or mechanisms are the "how" of achieving a function. This is known as the mechanistic answerers to "why red blood cells transport oxygen" is because oxygen binds to Hb molecules in RBCs.
Representative Functions
Exchange of oxygen and carbon dioxide between the internal and external environments
Perpetuation of the species
Coordination of body function through electrical signals and the release of regulatory molecules
Support and Movement
Protection from the external environment
Defense against foreign invaders
Coordination of body function through synthesis and release of reguatory molecules.
Conversion of Food into particles that can be transported into wastes
Transport of Materials between all cells of the body
Cardiac Muscle
Almost always cause an associated contraction. Cell membrane depolarization will periodically reach depolarization threshold and an action potential will fire, triggering contraction of the myocyte.
add single smooth unit action potentials
Action potentials (all or none) spikes.
Single-unit muscle cell membrane has voltage-gated channels, so can create an action potential when stimulated
Threshold potential is about -30mV
Relatively positive (perhaps -55 mV) due to high Na+ leak.
GRaph 2
Graph 1
Slow wave potential are unstable resting membrane potentials that continuously cycle through depolarization- and repolarization phases. However, not every cycle reaches depolarization threshold and thus an action potential (AP) will not always fir
Slow Wave Potentials:
Single-unit visceral smooth muscle is myogenic; it can contract regularly without input from a motor neuron .
Electrical Characteristics:
Lymphatic vessels
Smaller arteries / arterioles / smaller veins
Ureters and urinary bladder
the wall of the gastrointestinal tract
This organization is present in
Relatively sparse innervation
In this organization the smooth muscle cells are connected by gap junctions and there are fewer neurons..
Behaves in a syncytial manner.
Also called unitary or visceral smooth muscle.
add single unit smoothh muscle
Single-Unit Extreme end of Spectrum
phosphorylated cross bridges interact with actin to produce shortening
activated calmodulin activates the kinase enzyme
Ca2+ binds to calmodulin and activates it
Ca2+ interacts with calmodulin and myosin light chain kinase to activate myosin
Ca2+ is released from the SR and from the extracellular space
actin and myosin interact according to the sliding filament mechanism
add multiunit smooth muscle contraction mechanism
Contraction Mechanism
drugs
hormones
nerves
Tone may be modified by:
Tone is intrinsic property of the muscle itself -- does not depend on nerves (unlike skeletal muscle)
GI sphincters.
Example:
Constant and stable low level of contraction
Tone:
More stimulation causes graded-depolarization and graded response.
Multi-unit smooth muscle cell membrane lacks the voltage-gated channels that can cause action potentials.
add multiunity smooth muscles graded potentials
Multiunit Smooth Muscle uses graded potentials (PSPs)
Typically, do not display action potentials when stimulated to contract but displays graded potentials.
Membrane potential of multiunit smooth muscle is stable, and lacks many voltage-gated channels
Multiunit smooth muscle is neurogenic- that is, its contraction must be initiated by a motor neuron.
Electrical Characteristics
All sphincters of the body
Vas deferens
Iris Muscle
Bronchial muscles
Larger Blood Vessels
GI Sphincters
Tracheal Muscle
Ciliary muscles of the eye
These are found in places where fine control of contraction is needed
Higher innervation ratios than visceral smooth muscle.
Less gap junctions between cells
Each smooth muscle cell acts mostly independently (more like skeletal muscle).
Multi-Unit Extreme end of Spectrum
add multi-unit and single unit smooth muscle
Types of smooth muscle-
Sustained smooth muscle contractions are executed with minimal expenditure of ATP.
Slow cycling and long attachment of cross-bridges take place due to decrease in ATPase activity, and possibly a reduction in both MLCK and MLCP activity.
With latch state, ENERGY EXPENDITURE economy is high:
Contraction of SM is much slower than that of skeletal muscle. Force production can be equal to skeletal muscle.
Latch State
After reaching full contraction even if force of stimulus decreases the contraction is maintained for a long time
Latch mechanism:
Stress-relaxation: Hollow organs maintain same pressure despite changes in pressure.
Maximum force of contraction is more. 4-6kg/cm sq. Skeletal muscle about 3-4 kg.
Contraction time is 1 to 3 sec, about 30 times longer than in skeletal muscle.
Low energy requirement to sustain the same tension. About 1/10 to 1/300 that in skeletal muscles.
Slow cycling of actin and myosin cross bridges. About 1/10 to 1/300 that in skeletal muscles.
add comparison of smooth and skeletal muscle contraction
Comparison of SMooth and Skeletal Muscle Contraction
For relaxation of a sphincter (such as for urination) M3 receptors use their Gq protein to increase Ca++ in endothelial cells that produce NO.
The NO then diffuses into the sphincter muscle and causes it to relax.
Use Gq protein to increase intracellular Ca++ and cause smooth muscle contraction.
M3 receptors (and a smaller number of M1 receptors)
Use Gi proteins to inhibit adenylyl cyclase, lowering cAMP and thus causing smooth muscle contraction.
M2 receptors
Muscarinic Receptors
Ca++ channel blockers (L-type channels)
Ischemia (decrease in blood flow) opens ATP-sensitive K+ channels, hyperpolarization, closes Ca++ channels
Note: Both cAMP/PKA, and cGMP/PKG can phosphorylate phospholamban (PLB).
c GMP mediated response.
Nitric oxide causes smooth muscle relaxation in the same manner.
c AMP activates PKA which phosphorylates MLCK into a state where Ca+/calmodulin cannot bind.
High cAMP causes relaxation by effectively lowering myosin light chain kinase [MLCK]
Phospholamban (PLB) is an intracellular protein, when phosphorylated, it increases Ca++ re-uptake into SR by sarcoplasmic reticulum calcium pump (SERCA), clearing Ca++ from the cytosol.
Reuptake by the sarcoplasmic reticulum by SR.
Sarcolemma Ca++ATPase
Calcium extrusion from the cell eg. 3Na+/Ca++ exchanger
Low intracellular [Ca++] will cause the cell to relax.
Mechanism of Smooth Muscle Relaxation
High MLCK/MLCP Activity Ratio initiates smooth muscle contraction
Myosin light chain phosphatase (MLCP) is important for cessation of contraction i.e relaxation of muscle.
High Ca++ therefore causes contraction by increasing MLCK activity (increased in comparison with MLCP activity)
Calcium ions combine with calmodulin to cause activation of myosin light chain kinase (MLCK )and phosphorylation of myosin light chain (head)
Add mechanism of smooth muscle contraction
Mechanism of Smooth Muscle COntraction
This causes a neighboring group of cells to contract together. Smooth muscle cells contract in groups also because of gap junctions between cells that allow electrical signals to spread from one cell to another.
no specialized nerve cell junction
add smooth muscle varicosities
Neurotransmitter is released at varicosities (swellings) found along the length of the axon
Autonomic neurons do not form synapses with specific cells but with groups of cells.
Innervated by the autonomic nervous system.
Smooth Muscle Innervation
skeletal muscle (negligible-dependence).
cardiac muscle (partial-dependence)
Compare: Dependence on extracellular Ca++
Often needs additional extracellular Ca++ for activation and/or contraction.
Smooth muscle does have a SR containing Ca++, so Ca++ can be released from intracellular stores.
Poorly developed sarcoplasmic reticulum.
May involve graded potentials, that sometimes build into action potentials.
Smooth muscle does not always require action potential to contract.
Smooth muscle has no t-tubules, and a much less developed SR. Result is a slower, often less precise response
Small size of smooth muscle cells.
No T-tubules or terminal cistern system.
Lots of gap junctions in smooth muscle, but density varies from tissue to tissue.
Allow direct electrical communications between adjacent smooth muscle cells
GAP JUNCTIONS
Analogous to z-lines in striated muscle
Dense Bodies.
Serve as anchors for the thin-filament protein actin.
Cytoskeletal elements which form a structural backbone against which contraction occurs.
Intermediate Filaments
Lack neat hexagonal arrangement of actin and myosin
No troponin present at all
Therefore, Troponin-I does not inhibit cross-bridge cycling in smooth muscle as it does in skeletal muscle.
Exact functional role, if any, unclear.
Tropomyosin present in smooth muscle actin but:
F-Actin strands of G-Actin pearls
actin (thin-filament)
Location and action of the myosin light chains allows it to control activation of myosin cross-bridge heads.
So there are cross-bridge heads along the entire length of the myosin filament.
A main difference is the lack of a bare region in the middle lacking cross-bridges.
The organization of myosin molecules on the myosin filament is somewhat different than in skeletal muscle.
Light Chains - Play a key regulatory role
add myosin in smooth
myosin (thick, white filament).
add smooth muscle apparatus
Contractile Apparatus
The arrangement of dense bodies with connective-tissue fibers running throughout the cell and into adjacent gives structural integrity to the entire smooth muscle tissue, and anchor points for actin filaments
add smooth muscle architecture
Overall architecture
greater in smooth muscle (10:1) than in skeletal muscle.
Actin/myosin ratio:
Relatively small compared to skeletal and cardiac muscle
2-5 mm wide; 50-200 mm long.
Spindle shaped cells
Smooth Muscle
Skeletal and SMooth muscle
Write out steps from picture.
It tests L2, L3, and L4
This reflex is a reflex of proprioception which helps maintain posture and balance, allowing to keep one's balance with little effort or conscious thought.
otherwise its a monosynaptic reflex
This contraction, coordinated with the relaxation of the antagonistic flexor hamstring muscle causes the leg to kick.
From there, an alpha-motor neuron conducts an efferent impulse back to the quadriceps femoris muscle, triggering contraction.
Striking the patellar tendonwith a tendon hammer just below the patella stretches the muscle spindle in the quadriceps femoris muscle. This produces a signal which travels back to the spinal cord and synapses (without interneurons) at the level of L4 in the spinal cord, completely independent of higher centres.
Add patellar reflex
Patellar Reflex (GTOs)
this relaxes the tension on the muscle spindles, and reduces the discharge rate of the gamma efferents on the annulospiral endings.
As the skeletal muscles contract, the intrafusal muscles shorten;
During stretch, an action potential is transmitted to the central nervous system (CNS) at the spinal cord; the frequency of the action potential is proportional to the degree of stretching.
The stretch reflex is generated as a protective mechanism when the spindle and its skeletal muscles are being stretched.
Stretch Reflexes
Reflexes
Normal musccle fibers that surround the spindle are called extrafusal fibers.
don't stop sending signals
important in preventing muscle injury
Keep muscles stretched to keep signal going again and again to know how much load there is
to intrafusal muscle fibres
Gamma
to extrafusal (normal) muscle fibres
Alpha
Efferent
The branching end of the type II afferent fibers of these neurons are located on either side of the central bag region
Flower-spray endings
This is the end of a type Ia afferent fiber that wraps around the central bag region.
Annulospiral endings
There are two types of sensory receptors associated with the intrafusal fibers that detect the degree of stretch of these fibers which in turn monitors the degree of stretch of the entire muscle:
add muscle spindle 4
Afferent
add muscle spindle 3
Intrafusal Fibres
Diagrammatic cross-section of a muscle spindle showing
add muscle spindle 1 and 2
Muscle Spindle
Regulation of Muscular Contraction
This means that the actin and myosin fibers will remain linked until the muscles themselves start to decompose.
ATP reserves are quickly exhausted from the muscle contraction and other cellular processes.
Muscle stiffen after death.
Rigor mortis:
Eventually causes death because of paralysis of respiratory muscles
Recessive X –linked mutation in dystrophin gene in Duchenne muscular dystrophy.
Muscle dystrophy :
Needs the signals that maintain muscle tone to keep the muscle at a normal size.
Happens very quickly in cases of loss of nerve supply.
Happens more quickly when muscle is prevented from moving as in a cast following injury.
Occurs at approximately the same rate as hypertrophy during periods of non-exercise.
Muscle atrophy
More sarcomeres added to increase length if muscle is repeatedly over-stretched.
Increase in enzymes for glycolysis and in the number and size of mitochondria.
Increase in number of contractile proteins causing an increase in size of myofibrils up to 50%.
Hypertrophy (fibers)
States of Muscle Tone
Muscle blood flow may increase more than 20 fold during exercise
the increase is due to vasodilation and increase BP
resting = 3-6 ml/100g/min
max work = 90 ml/100g/min
add muscle blood flow in exercise
Muscle blood flow during exercise
far less ATP produced
temporarily maintains glycolysis without O2
Fermentation- lactate from pyruvate
At peak activity, anaerobic glycolysis needed to generate ATP:
oxidative phosphorylation in mitochondria
Glycolysis in cytoplasm;
Aerobic metabolism (requires O2) provides most ATP needed for contraction:
excess in blood with muscle damage.
Returns energy to ATP via enzyme creatine phosphokinase (CPK)
Creatine phosphate (CP) releases stored energy to convert ADP to ATP:
The creatine phosphate reserves of the muscle fiber can sustain contraction for about 5 to 8 seconds.
Creatine phosphate + ADP-------------------- > creatine + ATP
However, the ATP stores in the muscle can sustain muscle contraction for up to 3 seconds.
Thereafter, ATP is regenerated using the energy released by the dephosphorylation of creatine phosphate reserves of the muscle fiber.
In about 3 sec, all the ATP is depleted from the muscle cell.
ATP is the immediate source of energy for muscle contraction.
addd phosphagen system
Phosphagen System
insert energetics of muscle contraction
Energetics of Muscle Contraction
Fast-twitch fibers also have higher maximum shortening velocities compared to slow-twitch.
E.g. extraocular muscles
Some contain predominantly fast-twitch fibers (e.g. extraocular) and contract quickly.
Can mimic actions of FTG or STO fibers in response to specific muscle training.
crossover fiber.
Oxidative Glycolytic
Low endurance
High strength
Contains "fast myosin" which is able to hydrolyze ATP at a faster rate.
Few mitochondria.
Lots of constituents and enzymes for glycolysis.
Extensive SR.
Lots of contractile protein (large fiber size).
White in color, lacking much vascularization.
Glycolytic
Fast Twitch
high endurance.
Low strength,
Lots of mitochondria.
Highly vascularized.
Red in color.
Certain muscles (e.g. soleus) contain mostly slow twitch fibers and will contract slowly.
Slow Twitch Oxidative (aerobic)
This is because there are fast-twitch and slow-twitch fibers.
ADD tension-time graph for different muscles
Different types of fibers differ in their velocity of shortening
add velocity of shortening
When the velocity of shortening is plotted as a function of load, as the load increases the velocity of shortening gradually decreases
Shortest duration of contraction
Lowest velocity of shortening
Heavy load
latent phase is increased the most.
Longest duration of contraction
highest velocity of shortening
Zero load -
latent phase is shortest;
preload - isotonic - isotonic - isometric (rt. side = afterload)
add distance shortening - time
Graph of velocity against load
The duration of shortening decreases.
The velocity of shortening decreases
The latent period (time lag between stimulation and shortening) increases.
When a muscle contracts isotonically under increasing loads the contractions display the following effects:
Velocity of Shortening
Isometric Contractions occur when afterload is too heavy to lift
Afterload also determines how fast a muscle contracts during a lift.
Minimal loads allow the muscle to contract at maximal velocity.
the load the muscle is working against or trying to move during stimulation.
Afterload
Too much - you will fall on the right side of the curve
Too little - you will fall on the left side of the curve.
Stretching a muscle to optimize actin and myosin filament interaction is known as Preloading.
is the load on a muscle in a relaxed state, prior to contraction.
Preload
Preload is blood coming to hear to stretch it; afterload is blood in aorta and pushing it out into the vascular system
when you actually bend and lift - afterload
Example - outstretched arm picking up bucket of water - hold it straight - preload - stretching sarcomeres to optimal length
Preload and Afterload
Then as the sarcomeres get shorter the thick filaments come into contact with the Z lines and the decrease in tension with decreasing length becomes even steeper.
This results in a slow decrease in tension as the sarcomeres get shorter.
As the muscle becomes shorter than the optimum length the thin filaments at opposite ends of the sarcomere first begin to overlap one another and interfere with each other's movements.
When the muscle is stretched beyond this length the number of active cross bridges decreases because the overlap between the actin and myosin fibers decrease.
When the muscle is at the optimum length the number of active cross bridges is the greatest.
Factors Affecting the Force Generation of Individual Muscle Fibers
Changes in Fiber Length
Add changes in fibre length
Fiber Diameter
The greater the cross-sectional area of a fiber, the more force it can generate.
The greater the number of sarcomeres arranged in parallel, the greater the force generating capacity.
The number of sarcomeres arranged in parallel correlates with a fiber's diameter.
Frequency of Stimulation
add frequency of stimulation
When the frequency of stimulation is so high that Ca++ levels rise to peak levels, summation results in the level of tension reaching a plateau called tetanus.
At this point the tension plateau smoothes out and tetanus is called complete or fused.
At greater frequencies of stimulus, levels of Ca++ peak and cause a maximum number of crossbridges to cycle.
When the frequency of stimuli is high enough to cause tetanus but tension oscillates around an average level, the tetanus is called incomplete or unfused.
Increasing action potential frequency increases force by SUMMATION.
As muscle twitch is fairly slow compared to an action potential many action potentials can arrive before a single twitch is completed. This causes the twitches to bunch up and results in the generation of a force that is greater than a single twitch. This process is called summation.
Another possibility is that frequent stimulation "warms up" the muscle and thereby increases the enzymatic rate.
When a muscle is stimulated at a frequency so that twitches follow one another closely, the peak in tension rises in a step-wise fashion.
add frequency of stimulation 2
This phenomenon is called treppe.
add freq of stim 1(switch 3 somewhere else)
This in turn increases the number of crossbridges that form in the following contractions.
Treppe may occur because Ca++ released from previous twitches exceeds Ca++ reuptake and this results in an increase in Ca++ concentration.
The motor unit consists of a motor neuron and all the muscle fibers it innervates.
insert motor unit
Mechanisms of Skeletal Muscle Contraction
Types of Twitch
Relationship between Isotonic and Isometric Contraction in Real Life
add isometric vs. isotonic
The greater the load the higher the plateau and the greater the time lag between stimuli and the start of muscle shortening.
When the load exceeds the amount of force the muscle can generate, an isometric twitch results which is always of the same size and shape.
Isotonic Twitch
The tension curve for an isotonic twitch shows a plateau during which the force or tension is constant (iso-same; tonic- tension).
When the force of contraction of the muscle is at least equal to the load so that the muscle shortens, the muscle is said to contract isotonically.
An isotonic twitch is measured by attaching the muscle to a moveable load.
add isotonic twitch
Isometric Twitch
The rise and fall of tension traces a bell-shaped curve.
add isometric twitch
An isometric twitch is measured by keeping the muscle immobile while stimulating it and measuring the tension that develops during contraction.
When the load (force opposing contraction) is greater than the force of contraction of the muscle, the muscle creates tension when it contracts but does not shorten.
Phases of the the Twitch
When a stimulus is applied and a fiber contracts the twitch can be divided into three phases
add picture of phases
Relaxation
is the time between peak tension and the end of the contraction when the tension returns to zero. During this time cytosolic calcium is decreasing as reuptake exceeds release.
starts at the end of the latent period and ends when the muscle tension peaks (tension = force expressed in grams). During this time cytosolic calcium levels are increasing as released calcium exceeds uptake.
Latent
is the delay of a few milliseconds between an action potential and the start of a contraction and reflects the time for excitation-contraction coupling.
Ca build up?
A twitch is the mechanical response of an individual muscle fiber, an individual motor unit, or a whole muscle to a single action potential.
Muscle contraction is based on a twitch of the muscle fiber which is like an action potential is an all or nothing event.
Removal of Ca++ from the sarcoplasm (relaxation)
Large additional stores of un-ionized calcium is bound in SR by a protein called calsequestrin. Will be released as ionized Ca++ to maintain stores in cisternae if they get low.
SR is very effective storage unit for calcium and calcium ions. Pump actively moves ions into cisternae.
Calcium pump actively transports Ca++ out of the sarcoplasm to end the contraction. Very efficient in clearing the intracellular fluid almost entirely of Ca++.
Excitation Contraction Coupling
Add picture - excitation contraction coupling
Simply means the process that excites the cell membrane at the neuromuscular junction is coupled to the processes that cause muscle contraction.
Release of Ach at the neuromuscular Junction
lots of ACh receptors located near mouths of subneural clefts.
Post-synaptic membrane receives signal
Diffuses across to post-synaptic side.
Exocytosis releases ACh into cleft.
Bound to membrane with docking proteins.
Terminal vesicles containing ACh move to the membrane
Voltage gated Ca++ channels open.
Depolarization reaches axon terminal.
The Motor End Plate
Insert Picture - Motor End PLate
Picture Captions
Folds in the bottom of the trough, “subneural clefts”, to increase surface area for stimulation by Ach.
Acetylcholinesterase in synaptic cleft to lyse and clear ACh from cleft when signal ends.
Invagination of post-synaptic membrane called a “trough” or “gutter”. 20-30nm wide.
Lots of mitochondria in neuron terminal to provide ATP energy for synthesis of ACh.
Typically near center of muscle fiber. Extremely long fibers may have two neuromuscular junctions.
Branch to stimulate 3 – 100s of muscle fibers at motor end plates (neuromuscular junction). Each branch is myelinated by schwann cells.
Innervated by myelinated somatic neurons (alpha)
Length-Tension Relationship Sarcomeres
When the filaments are pulled too far from one another, as seen at point 4, they no longer interact and cross-bridges fail to form. Zero tension results
Maximum tension is produced when sarcomeres are about 2.1 to 2.2 μm long, as seen at point 2. This is the optimal resting length for producing the maximal tension.At point 3, there is little interaction between the filaments. Very few cross-bridges can form. Less tension is produced.
At point 1 in Graph, the sarcomere is overly contracted. There is a high degree of overlap between the thin and thick filaments. Muscle contraction causes actin filaments to slide over one another and the ends of myosin filaments. Further muscular contraction is halted by the butting of myosin filaments against the Z-discs. Tension decreases due to this pause in cross-bridge cycling and formation.
Changes in Resting vs. Active Sarcomere
Sliding Filament Theory
Contraction
Continued Contraction
As long as Ca++ remains in sarcoplasm, actin sites remain exposed, and ATP is available, the heads will recycle rapidly repeatedly pulling actin past their position.
“Walk-Along or “Ratchet” theory
New ATP splits and the released energy “cocks” head again for next stroke.
New ATP then binds head, causing release from actin.
ADP and Pi leave the myosin head triggering Power Stroke.
Myosin cross bridge binds to site on actin filament, creating ACTOMYOSIN.
Insert ACtin-Myosin Interaction picture
ATP in myosin cross bridge head already cleaved to ADP + Pi by ATPase in head but all are still bound to head. It puts arm and head into “cocked” position.
Ca++ binds with troponin (on the actin filament) causing conformational change that removes inhibition from binding site.
Excitation
Ca2+ then bind to troponin and activates the contraction process (Jurkat-Rott and Lehmann-Horn 2005)
activates the ryanodine receptor (RyR) that releases Ca2+ from the SR.
A receptor protein dihydropyridine receptor (DHP) senses the membrane depolarization, alters its conformation, and
A muscle fiber is excited via a motor nerve that generates an action potential that spreads along the surface membrane (sarcolemma) and the transverse tubular system into the deeper parts of the muscle fiber.
Ca++ floods the muscle cell
Ca++ receptors along the SR open even more Ca++ channels
Change in voltage of SR induces release of Ca++ from cisternae (voltage-gated Ca++ channels in SR)
Signal moves down t-tubules to SR
Depolarization of sarcolemma from center toward ends of cell
Signal at neuromuscular junction (motor end plate)
Sarcoplasmic Reticulum = Endoplasmic Reticulum
Add in picture
Rough
Constant replacement / synthesis of contractile proteins.
Protein synthesis.
Covered with ribosomes.
T-Tubules
Mammalian skeletal muscle has two T tubules per sarcomere.
Where t-tubules contact terminal cisternae. Forms a "triad" including t-tubule with cisternae on both sides.
They branch when inside the cell forming extensive "plates" for efficient delivery of the electrical signal.
Transverse (t) tubules carry electrical signal from sarcolemma into fiber.
Smooth SR surrounds each myofibril
Can release or clear sarcoplasm of Ca++ ions (clears Ca++ via active transport).
Extensive network for storage of Ca++ ions in terminal cisternae.
Sarcoplasm = Cytoplasm
Contains normal organelles and cytoskeleton
The Physiological Components of the Body
Physiology is a special focus on the integration of function among systems across many levels of human body organization.
The figure below is a “map” of how all the physiological components of the body are linked together to maintain life.
Maps in physiology usually focus either on the relationships between anatomical structures and physiological processes or on normal homeostatic control pathways and responses to abnormal events. Mapping is a way of organizing material to help study the importance of and relationships between various pieces of information.
Musculoskeletal System
Muscle Physiology
Types of Muscles
Smooth
Striated
Cardiac
Skeletal
Structure of Skeletal Muscle
Muscle ensheathed by Epimysium
Note that the sheaths travel length of muscle to form tendons at each end
Composed of Fascicles ensheathed by Perimysium
Composed of Myofibres ensheathed by Epimysium
Also known as muscle cell or myocyte
Composed of 100s - 1000s of Myofibrils
Composed of very organized system of Myofilaments
Organized functionally into sarcomeres
Overall light/dark areas depend on density of fibers.
Bare Zone
As actin fibers move close together and enter this zone, they cannot produce additional force.
A small area close to the M-line contains no crossbridge heads.
M Line
Z line
Actin filaments pierce Z line and have an end in each of two sarcomeres.
They are anchored here
(disc) separates sarcomeres.
H zone
in center contains only myosin
A band
dark) contains both actin and myosin.
I Band
(light) contains only actin at rest.
Regular organization with six actin filaments surrounding each myosin filament.
Shared by adjacent myosin filaments
Might be considered the functional units of the muscle.
Composed of fibres/proteins
Ancillary Proteins
Tropomyosing
Troponin Complex
Titin
Holds microfilaments in place and provides elastic recoil.
Very flexible and springy.
filaments (MW of 3,000,000).
Nebulin
Primary Contractile Fibres
Myosin
Hinges at two places
Arm
Head
200 Myosin Molecules
arranged with tails bunched together so that arms & heads are free to swivel
Heads face out in all directions.
Each molecule is composed of
head
Important characteristics
it has ATP-binding sites into which fit molecules of ATP
Subcomponents
Light Chain Molecules
Heavy Chain Molecules
heavy chain tail,
1500 per myofibril
Actin
Troponin
attached to tropomyosin has strong affinity for
Ca++
When Ca++ influx occurs and Ca++ binds with troponin, it pulls on tropomyosin causing a conformational change that exposes actin binding sites.
Tropomyosin
Tropomyosin
Cover active sites on G-actin molecules preventing attachment of myosin cross bridges.
Function is to keep active sites covered.
wrap around F-actin helix.
fiber like” molecule 50nm long
Double helix of F-actin protein (backbone)
made of G-actin molecules .
Each G-actin molecule has one ACTIVE SITE where ADP is attached.
Each bead is a G-actin.
Each actin is one micrometer long, anchored in Z line
3000 per myofibril
About twice as many actin filaments as myosin filaments
Contractile Filaments
ER: Extensive sarcoplasmic reticulum
Mitochondria: Many
Nucleus: Multi-nucleated
Plasma Membrane: Sarcolemma
Diameter: 10-80 micrometers
Length: Can be very long (length of a muscle)
Intgumentary System
Urinary System
Kidneys
Bladder
Reproductive System
Uterus
Ovaries
Testes
Endocrine System
Thyroid Gland
Nervius System
Brain
Immune System
Lymph Nodes
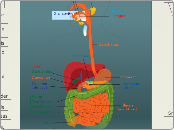
Digestive System
Intestines
Stomach
Mouth
Cardiovascular System
Heart
Blood Vessels
respiratory System
Lungs
Basic Biological Concepts Involved with Maintaining Human Life
Communication
Communication between both structure and function (integration) is required for cells in the body to communicate rapidly and efficiently
Mass Flow
mass flow refers to the movement of substances within and between compartments of the body
- the movement of materials in mass flow is made possible by a driving force such as a pressure or concentration gradient
Information Flow
information flow in the body takes the form of either chemical signals or electrical signals
information may pass from cell to its neighbors (local communication) or from one part of the body to another (long-distance communication)
information stored in the genetic code of DNA is passed from one cell to its daughter and from an organism to its offspring
Distance Travelled
Short Distance
i.e. from a cell to its neighbours
Long Distance
i.e. from one body part to another
Information Type
Electrical
Chemical
Structural-Functional Relationships
. structural-functional relationships demonstrates the molecular interactions, the division of the body into discrete compartments, and the mechanical properties of cells, tissues, and organs
Molecular Interactions
Mechanical Properties of Cells, Tissues and Organs
Compartmentalization
Biological Energy Use
biological energy use involves the synthesis and breakdown of molecules to provide energy used to transport substances across cell membranes and to create movement
Homeostasis
homeostasis is defined as the maintenance of a relatively stable internal environment which is maintained by monitoring and adjustments by physiological control systems
Types of Control Loops
Non-Homeostatic - Positive Feedback System
. in a positive feedback system, the reflex response to the disturbance will be the same as the disturbance
• this type of control is mostly used for intensifying a change, not to correct it
An outside factor will be required to shut of the positive feedback loop
• examples -
the propagation of an action potential down a cell membrane;
oxytocin increases uterine smooth muscle contractions which stimulate more oxytocin
secretion;
blood clotting cascade
A few reflex pathways are not homeostatic. In a positive feedback control system, a variable is sensed and action is taken to reinforce the change of the variable. Since the response is in the same direction as the disturbance, there is a cumulative or amplified effect associated with the reflex. Positive feedback does not lead to stability or regulation, but to the opposite - a progressive change in one direction. This type of control is mostly used for intensifying a change, not to correct it.
The small figure in the upper right shows that A stimulating B results in B
stimulating A.
Pathological Effects
The above diagram shows how a positive feedback mechanism leads to the loss of homeostasis.
It shows that as the abdominal cavity fills with fluid leaked from injured organs, the increased pressure impairs blood flow and leads to cell death.
This initiates a cascade of positive feedback events that can eventually lead to death of the patient.
Sudden loss of 2 L of blood from the body leads to a condition where heart muscle will not get enough blood for normal functioning.
Thus, a weakened heart will pump less blood to heart muscle which further reduces the efficiency of the heart. These cyclical processes continue and death will eventually occur.
Other Examples
the blood clotting cascade
the propagation of an action potential
the calcium-induced calcium ion release in cardiac cells during each heart beat
during the follicular phase of the menstrual cycle, estrogen stimulates the release of luteinizing hormone which in turn stimulates further estrogen synthesis by the ovaries
Initiation of Urination when Bladder is full
Parturition
An example of a positive feedback loop involves the hormonal control of uterine contractions during childbirth (figure on the right).
When a baby is ready to be delivered, it drops lower in the uterus and begins to put pressure on the cervix.
Sensory signals from the cervix to the brain cause release of the hormone oxytocin,
which causes the uterus to contract and push the baby’s head even harder against the cervix, further stretching it.
The increased stretch causes more oxytocin release, which causes more contractions that push the baby harder against the cervix.
This cycle continues until the baby is delivered, ending the stretch on the cervix and stopping the positive feedback loop.
Negative Feedback System
the reflex responses to the disturbance will be
opposite to the disturbance (by far the most common physiological mechanism used)
• examples - if arterial blood pressure decreases, the reflex response will be to increase the pressure; if plasma sodium decreases, the reflex response is
to increase plasma sodium
If the response loop is successful, it shuts itself off.
In a negative-feedback control system, information flows along a closed loop.
The regulated variable is sensed and information about its level is fed back to a feedback controller, which compares it with the desired value (set point).
If there is a difference, an error signal is generated, which drives the effector to bring the regulated variable closer to the normal value.
The reflex response is opposite to the disturbance = negative feedback.
Arterial Baroreceptor Reflex
Cardiovascular Response to Hemorrhage
Hemorrhage will stimulate both the arterial baroreceptors and the stretch receptors of the renal afferent artery.
Arterial blood pressure receptors are responsible for acute reflex (nervous reflexes) responses to a decrease in blood pressure while the renal system is responsible for chronic reflex (hormonal) control to reestablish arterial blood pressure. Increasing TPR will result in net absorption in the capillaries to increase intravascular volume.
Mean Arterial Blood Pressure
Subtopic
The neural reflex system that regulates mean arterial blood pressure (MAP) operates as a series of negative feedback loops. A dual system of sensors and neural reflexes controls mean arterial blood pressure:
1. the primary sensors are baroreceptors which are actually stretch receptors or
mechano-receptors that detect distension of the vascular wall
• arterial blood pressure detectors are located in the vascular walls of the carotid
sinus and in the muscular walls of the aortic arch
2. the secondary sensors are chemoreceptors that detect changes in blood PO2, PCO2,
and pH
The control centers are located within the CNS, mostly in the medulla, but sites within the cerebral cortex and hypothalamus also exert control. The effectors include the pacemaker and contractile muscle cells in the heart, the vascular smooth muscle cells in arteries and veins, and the adrenal medulla. The entire control process can be referred to as the baroreceptor reflex control of arterial blood pressure and consists of the:
1. arterial baroreceptors = the detectors (sensors)
2. afferent neural pathways = the input pathways
3. control centers in the medulla = the integrating centers
4. efferent neural pathways = the output pathways
5. heart and blood vessels = the target tissues
Gain
GAIN = CORRECTION RESIDUAL ERROR
-the less variation you can tolerate, the greater the gain (eg. body temp has gain of 27, blood pressure is 2) (i.e. can tolerate great change in blood pressure without dying, but can’t tolerate great change in body temperature)
The degree of effectiveness with which a control system maintains a constant condition is determined by the gain of the negative feedback control system.
The efficiency of a control system is related to the range within which the controlled variable must be maintained.
The smaller the normal range of a given physiological variable, the higher the gain must be and therefore a more efficient control system to maintain the variable within the normal limits (or the established set point). Some examples are:
1. body temperature control system gain averages about 27
2. baroreceptor arterial blood pressure control system <2
3. renal arterial blood pressure control system is infinite
Set point is defined as the normal value a control system attempts to maintain.
As illustrated in the previous page, there may be many control systems with unequal gains involved with maintaining a given physiological variable. All the control systems shown, plus others, are required for maintaining arterial blood pressure and preserve homeostasis so life can continue.
Feedforward Control
Negative feedback loops can stabilize a function and maintain it within a normal range but are unable to prevent the change that triggered the reflex.
A few reflexes have evolved that enable the body to predict that a change is about to occur and start the response loop in anticipation of the change. These anticipatory responses are called feedforward control.
An example of feedforward control is how the sight, smell, or even the thought of food can stimulate the secretion of saliva in the oral cavity and hydrochloric acid in the stomach.
Local Control: Isolated Changes
a relatively isolated change occurs in the vicinity of a cell or tissue and evokes a paracrine or autocrine response (discussed on next page)
• the response is restricted to the region where the change took place
an example:
cells lining small blood vessels (arterioles) can sense a decrease in oxygen concentration and respond by secreting a paracrine signal which causes:
smooth muscle in the blood vessel to relax (dilate) →
decreases vascular resistance → increases blood flow →
increases oxygen delivery
Signalling Systems Involved
Cell to Cell
cells may communicate with each other directly via gap junctions or chemical messengers
Paracrine
- a chemical messenger diffuses a short distance through the extracellular fluid and binds to a nearby cell
Autocrine
a chemical messenger diffuses a short distance through the extracellular fluid and binds to a receptor on the same cell
Long Distance Control via Reflex Responses: Systemic Changes
(more complicated reflex loops) respond to
changes that are more widespread throughout the body or systemic in nature
• an integrating center located away from the affected tissue receives information, evaluates it, and decides whether to send a chemical or electrical signal to initiate a response
• the long-distance reflex pathway normally involves two control systems:
- the nervous system, normally is a rapid response system
- the endocrine system, normally is a more prolonged effect because of increased
time in signaling and extended target tissue effect
Signalling System involved
Neuroendocrine
involves the release of a hormone from a nerve cell and the transport of the hormone by the blood to a distant target cell
Endocrine
normally is a more prolonged effect because of increased time in signaling and
extended target tissue effect
involves the release of a hormone into the bloodstream and the binding of the hormone to a specific target cell membrane
Nervous System
Rapid response, less prolonged.
- involves the rapid transmission of action potentials, often over long distances, and the release of a neurotransmitter at a synapse
Physiological Control Systems/Reflexive Pathways
Homeostasis is the ability of the body to maintain a relatively stable internal environment.
This is accomplished by a continuous process that uses a physiological control system (i.e. physiological response loop) to monitor key functions, which are often referred to as regulated variables.
In its simplest form, any control system has three basic parts: Input Signal, Controller, Outpurt SIgnal
Each reflex control mechanism has a different ability to return a disturbance to a physiological system to normal. As shown above, each control system regulating mean arterial blood pressure has its own time course of action and its own efficiency in returning a disturbance towards normal. Each system uses different physiological mechanisms to accomplish their goal.
Gain is how efficiently a control system can return a disturbance back to normal
(Regulated Variable)
As with any control system, the three components of a response loop can be broken down into seven steps to form a pattern that is associated with all reflex pathways (with slight variations):
stimulus → sensor or receptor → afferent pathway →
integrating center →
efferent pathway → target or effector → response
1. a stimulus is the disturbance or change that sets the pathway in motion
• the stimulus may be a change in temperature, arterial blood oxygen content,
plasma glucose levels, or arterial blood pressure
2. a sensor or sensory receptor continuously monitors its environment
3. an afferent signal links the receptor to the integration center (incoming sensory
signals)
4. the integration center evaluates the incoming signal and compares it
to the set point (or desired value) and decides the appropriate response
5. the integrating center then initiates the efferent pathway
• outgoing efferent signals may be either stimulatory or inhibitory in nature
6. an electrical or chemical signal then travels to the effector
7. the effector carries out the appropriate response to bring the situation
back to within normal limits
Within the desired range (homeostasis)
No Action Required
Outside of Desired Range
Away from set point
7. Response
the effector carries out the appropriate response to bring the situation back to within normal limits
Not Successful
If the body fails to maintain homeostasis, the normal function is disrupted and a pathological condition will exist. Diseases can be divided into two general groups according to their origin:
In both internally and externally caused diseases, when homeostasis is disrupted, the body attempts to compensate. If the compensation is successful, homeostasis is restored. If compensation fails, illness and disease may result. The study of body functions in a disease state is known as pathophysiology
External Failure
2. external causes of disease include toxic chemicals, physical trauma, and foreign
invaders such as viruses and bacteria
Internal Failure
. those in which the problem arises from internal failure of some normal physiological process
• examples would include cancer, autoimmune diseases, and inherited disorders
Successful
Example
Water temperature increases
Heater turns on
Signal Passes from wire to heater
Control box is programmed to response to a temperature below 29 degrees
Signal passes from centre to control box through wire
Thermometer senses the decrease
Water Temperature is below the set point
6. Effectors
Effectors are muscles or other tissues that influence that regulated variable
an electrical or chemical signal then travels to the effector
5. (Output Signal)/Efferent Pathway
. the integrating center then initiates the efferent pathway
originating from the integrating center is often from a neuron or an endocrine cells
outgoing efferent signals may be either stimulatory or inhibitory in nature
4. (Controller) Integration Centre
a controller acts as an integrating center that evaluates information coming from the sensor and initiates a response that is designed to bring the regulated variable back into the normal range
4. the integration center evaluates the incoming signal and compares it
to the set point (or desired value) and decides the appropriate response
3. (Input Signal) Afferent Signal
1. an input signal consists of
the regulated variable and
a specialized sensor
• if the variable moves out of its programmed range, the sensor is activated and sends signals to the controller
an afferent signal links the receptor to the integration center (incoming sensory signals)
2. Sensor
. a sensor or sensory receptor continuously monitors its environment
1. Stimulus
. a stimulus is the disturbance or change that sets the pathway in motion
Example: the stimulus may be a change in
temperature,
arterial blood oxygen content,
plasma glucose levels, or
arterial blood pressure
Environmental Conditions Required for Life
Cellular Physiology and The Body Fluid Compartments
Cellular Membranes
Membrane Potentials
ACTION POTENTIALS AND THEIR CONDUCTION
THE EFFECTS OF STIMULATION ON THE MEMBRANE POTENTIAL OF AN EXCITABLE CELL
Insert THE EFFECTS OF STIMULATION ON THE MEMBRANE POTENTIAL OF AN EXCITABLE CELL
The effects of successively applied stimuli of progressive strength are shown above
at point “D”, the stimulus is now strong enough to increase the acute local potential change to rise to a threshold level and generate an action potential
an action potential is defined as an electrical signal that propagates over a long distance without a change in magnitude
AXON DIAMETER CORRELATES TO THE SPEED OFACTION POTENTIAL CONDUCTION
The speed with which action potentials propagate depends on two key physical parameters:
Myelination
DEMYELINATING DISEASES AFFECT CONDUCTION VELOCITY
Most demyelinating diseases are either inherited or autoimmune disorders and result in the deterioration of the myelin sheath
These diseases are characterized by a variety of neurological complaints such as:
In demyelinating diseases, the loss of myelin from vertebrate neurons can have devastating effects on neural signaling.
In addition, when ions leak out of the now-uninsulated regions of membrane, between the channel-rich nodes of Ranvier, the depolarization that reaches a node may not be above threshold and conduction may fail.
In the central and peripheral nervous systems, the loss of myelin slows the conduction velocity of action potentials
The normal saltatory conduction of an action potential down a myelinated axon is illustrated in this figure.
MODES OF ACTION POTENTIAL CONDUCTION
The above figure summarizes the modes of action potential conduction in unmyelinated and myelinated axons:
figure “C” shows the propagation of an action potential in a myelinated
jumping from one node to the next is referred to as Saltatory Conduction
the initiation of an action potential in one node of Ranvier depolarizes the next node
figure “B” shows a sheath of myelin surrounding an axon
Figure “A” shows the propagation of an action potential in an unmyelinated axon
by sequentially depolarizing adjacent segments of the axon, the action potential propagates or moves along the length of the axon from point to point, like a traveling wave
SCHWANN CELLS FORM MYELIN AROUND PERIPHERAL NERVES
This myelin sheath insulates the axon and prevents the passage of ions through the axonal membrane
creates a high-resistance wall
Myelin forms when these glial cells wrap around an axon, squeezing out the glial cytoplasm so that each wrap becomes two membrane layers
They wrap themselves around axons to form myelin, a substance composed of multiple concentric layers of phospholipid.
Two different cell types in the CNS produce myelin
oligodendrocytes in the central nervous system (CNS)
Schwann cells in the peripheral nervous system (PNS)
Each Schwann cell wraps around a 1.0 - 1.5 mm segment of the axon leaving tiny gaps
nodes of Ranvier
therefore the node plays an important role in the transmission of electrical signals along the axon.
SALTATORY CONDUCTION
The reason that conduction velocity is more rapid in myelinated axons is because the mechanical process of ion channel opening slows conduction slightly.
In myelinated axons, only the nodes need Na+ channels because of the insulating properties of the myelin sheath.
As the action potential passes along myelinated segments, conduction is not slowed by channel opening.
In unmyelinated axons, channels must open all the way down the axon membrane to maintain the amplitude of the action potential.
The conduction velocity in mammals ranges from 3 to 120 m/sec for myelinated axons and 0.5 to 2.0 m/sec for unmyelinated axons.
This process results in a faster conduction velocity for myelinated than for unmyelinated axons
Action potentials are successively generated at neighboring nodes of Ranvier, therefore the action potential in a myelinated axon appears to jump from one node to the next
a process called saltatory conduction
When an action potential is initiated, the influx of Na+ causes the adjacent node of Ranvier to depolarize, resulting in an action potential at the node.
In myelinated axons, voltage-gated Na+ channels are highly concentrated in the nodes of Ranvier where the myelin sheath is absent and are in low density beneath the segments of myelin.
At each node a tiny section of axon remains in direct contact with the extracellular fluid and has ion channels through which current can leak
The second key parameter that is involved with determining the conduction velocity of an action potential traveling down a membrane is myelin
Diameter
the speed at which an action potentials are conducted, or conduction velocity, increases as a function of increasing axon diameter and concomitant increase in the space constant
this increase in ion flow in the cytoplasm causes greater lengths of the axon to be depolarized, decreasing the time needed for the action potential to travel the length of the axon
the space constant (λ) determines the length along the axon that a voltage change is observed after a local stimulus is applied in this case, the local stimulus is the inward sodium current that accompanies the action potential
the larger the space constant, the farther along the membrane a voltage change is observed after a local stimulus is applied
will influence the speed of propagation because of a decrease in the resistance to electrical current flow
diameter pipe because of their decreased resistance, large-diameter axons have less cytoplasmic resistance, thereby permitting a greater flow of ions
the diameter of the axon will influence the speed of propagation because of a decrease in the resistance to electrical current flow
After an action potential is generated, it propagates along the axon toward the axon terminal:
: it is conducted along the axon with no decrement in amplitude.
CONDUCTION OF AN ACTION POTENTIAL
Mechanism
THE GENERATION OF ACTION POTENTIALS ALONG AN AXON
As the first domino falls, it strikes the next, passing on its kinetic energy to the second domino which then falls, and so on.
The action potential that occurs at the trigger zone (or motor endplate) is like the movement in the first domino of a series of dominos standing on end (top figure).
When talking about action potentials, it is important to realize that there is no single action potential that moves through the cell.
The initiation and the sequential propagation of an action potential down an axon or across a muscle fiber is illustrated above
the section of cell membrane proximal to the active area is in an absolute refractory period
therefore the direction of propagation is in one direction
charge of the resting membrane potential
the voltage-gated Na+ channels on the cell membrane distal to the depolarized region begin to open, allowing Na+ to enter into the cell
at the peak of each action potential, the Na+ channels become inactivated and K+ channels open allowing K+ to exit the cell initiating repolarization
continuous entry of Na+ down the axon toward the axon terminal (or muscle fiber) means that the strength of the signal does not diminish as the action potential propagates itself (compare this to a graded potential)
this starts the positive feedback loop:
causing more depolarization and opening more Na+ channels in the adjacent membrane
Na+ enters
depolarization opens Na+ channels
shows that a positive charge from the depolarized region spreads to adjacent sections of the membrane
repelled by the Na+ that entered the cytoplasm and attracted by the negative charge of the resting membrane potential
shows that depolarization opens voltage-gated Na+ channels and Na+ enters the cell and the local area depolarizes (trigger zone or motor endplate)
” a stimulus that is a graded potential above threshold that enters the trigger zone an axon or is generated at the motor endplate of the neuromuscular junction
Whenever depolarization reaches those channels, they open, allowing more Na+ to enter the cell reinforcing the depolarization.
This process initiates the positive feedback loop or the propagation of the action potential
The axon or muscle cell membrane is well supplied with voltage-gated Na+ channels.
The current flow in the cytoplasm diminishes over distance as energy dissipates, and would eventually die out if not for voltage-gated channels.
Simultaneously, on the outside of the cell membranes, current flows back toward the depolarized section.
At the cellular level, the depolarization of a section of an axon or muscle fiber causes positive current to spread through the cytoplasm in all directions by local current flow, as illustrated in the figure above.
The action potential that reaches the end of the axon or muscle fiber is identical to the action potential that started at the trigger zone on an axon or the site of stimulation on a muscle fiber.
In action potentials, the flow of electrical energy is a process that constantly replenishes lost energy, which is why an action potential does not lose strength over distance as a graded potential does.
The high speed movement of an action potential along the axon or muscle fiber is called conduction of the action potential.
GRADED POTENTIALS DECREASE IN STRENGTH AS THEY SPREAD OUT FROM POINT OF ORIGIN
Graded potentials in neurons are depolarizations that occur in the dendrites and cell body or, less frequently near the axon terminals.
THE FREQUENCY OF ACTION POTENTIAL FIRING INDICATES THE STRENGTH OF A STIMULUS
A graded potential reaching the trigger zone does not usually trigger a single action potential, instead it triggers a burst of action potentials (figure b)
The transmission of information is not dependent on the strength and/or duration of the stimulus,
therefore it is not the amplitude of the action potential, but the frequency of the action potentials that results in transmitting the desired information.
One distinguishing characteristic of action potentials is that every action potential in a given neuron or muscle fiber is identical to every other action potential in that neuron or muscle.
A COMPARISON OF GRADED POTENTIALSTO ACTION POTENTIALS
GRADED STRENGTH OF A STIMULUS
In muscle cells, the graded potential occurs at the neuromuscular junction and is referred to as the End Plate Potential (EPP), this will be discussed in more detail in the neuromuscular junction lecture.
Although this graded potential also diminishes with distance through the neuron, its higher initial strength ensures that it is above threshold at the trigger zone.
A stronger initial that initiates a stronger depolarization and ultimately causes an action potential stimulus (figure b).
As a result the graded potential is below threshold by the time it reaches the trigger zone (figure a).
Although the cell is depolarized to -40 mV at the site where the graded potential begins, the current decreases as it travels through the cell body.
A single stimulus triggers a subthreshold graded potential, one that is below threshold by the time it reaches the trigger zone.
Two neurons with three recording electrodes placed at intervals along the cell body and trigger zone are shown in this figure.
The reasons that graded potentials lose strength as they move through the cytoplasm is because of two reasons:
cytoplasmic resistance
to the electrical flow is provided by the cytoplasm itself, just like water creates resistance that diminishes the wave from the stone
current leak
due to some of the positive ions leaking back across the membrane due to some of the positive ions leaking back across the membrane
the membrane in the neuron cell body is not a good insulator and has open leak the membrane in the neuron cell body is not a good insulator and has open leak channels that allow positive charge to flow out of the cell
The stronger the initial amplitude, the farther the graded potential can spread through the neuron before dying out.
If more Na+ channels open, more Na+ enters, and the graded potential has a higher initial amplitude.
By convention, current in biological systems is the net movement of positive electrical charge.
The positive charge carried by the Na+ spreads as a wave of depolarization through the cytoplasm, just as a stone thrown into water creates ripples or waves that spread outward from the point of entry.
The wave of depolarization that moves through the cell is known as local current flow.
A graded potential that begins when a stimulus opens monovalent cation channels on the cell body of a neuron. Sodium ions move into the neuron, bringing in electrical energy.
A large stimulus causes a strong graded potential, and a weak stimulus results in a weak graded potential.
These changes in membrane potential are called “graded” because their size, or amplitude is directly proportional to the strength of the triggering event.
VARIOUS CHEMICALS CAN INACTIVATE THE VOLTAGE-GATED Na+ AND K+ CHANNELS
Insert VARIOUS CHEMICALS CAN INACTIVATE THE VOLTAGE-GATED Na+ AND K+ CHANNELS
Tetra ethylammonium (TEA+)
blocks the voltage-gated K+ channels by entering the channel from the cytoplasmic side and completely blocks the membrane channel.
What will this do to further development of generating additional action potentials (propagation of an impulse)?
What effect will this have on the repolarization process?
Saxitoxin
What could be the possible causes of death?
Shellfish eat the dino-flagellates, and saxitoxin becomes concentrated in their tissue.
Saxitoxin is produced by red dinoflagellates that are responsible for the so-called “red tide”.
is another voltage-gated Na+ channel blocker
tetrodotoxin (TTX)
Sushi chefs are trained to remove the ovaries safely and are licensed by the government. Nevertheless, several people die each year from eating improperly prepared puffer fish.
What are the possible causes of death from ingesting TTX?
Raw puffer fish is a highly prized Japanese food dish. Connoisseurs of puffer fish enjoy the tingling numbness of the lips that is caused by very small amounts of TTX present in the flesh.
What are the possible causes for this sensation?
It binds to the extracellular side of the voltage-gated Na+ channels causing inactivation of the channel (blocks the activation gate), therefore no action potentials can be generated in nerve and/or muscle fibers.
The ovaries of certain species of puffer fish, also known as blowfish, contain tetrodotoxin (TTX) which is one of the most potent poisons known.
TYPICAL TRANSMEMBRANE ACTION POTENTIALSOF VARIOUS CARDIAC MUSCLE CELLS
Insert TYPICAL TRANSMEMBRANE ACTION POTENTIALSOF VARIOUS MUSCLE CELL TYPES FOUND WITHIN THE HEART
The transmembrane action potentials along with the correlation to the extracellular electrical activity (ECG) are shown in the figure above.
The structures that make up the conduction system are the
and the Purkinje system
the bundle of His and its branches
the atrioventricular node (AV node)
the internodal atrial pathways
sinoatrial node (SA node),
The heart beat originates in a specialized cardiac conduction system and spreads via this system to all parts of the myocardium.
The parts of the heart normally beat in an orderly sequence: contraction of the atria is followed by contraction of the ventricles, and during diastole all four chambers are relaxed.
ION EXCHANGE IN A CARDIAC MUSCLE CELL
Insert ION EXCHANGE IN A CARDIAC MUSCLE CELL
Note the complexity of ion exchange that is occurring during a ventricular electrical action potential. There is an incredible degree of balancing the chemical and electrical gradients for each ion to achieve the desired results.
ACTION POTENTIAL REFRACTORY PERIODS
Insert ACTION POTENTIAL REFRACTORY PERIODS
The explosive depolarizing phase of the action potential may be compared to a chemical explosion.
Voltage inactivation of Na+ channels partially accounts for important properties of excitable cells, such as the refractory periods.
Where more than one channel of a particular type is shown, the population is split between the states.
A single channel during a phase in the above figure means that the majority of channels are in this state.
During the latter part of the action potential, the cell is able to fire a second action potential, but a stronger-than-normal stimulus is required
Throughout the relative refractory period, the conductance of K+ is elevated, which opposes depolarization of the membrane adding to the refractoriness.
The resetting of the inactivation gate determines when the absolute refractory period ends and the relative refractory period begins.
Early in the relative refractory period, before the membrane potential has returned to the resting potential level, some Na+ voltage-gated channels are becoming excitable while others remain inactivated thus requiring a stronger stimulus to open the critical number of Na+ channels needed to trigger an action potential (threshold)
this is called the relative refractory period
During much of the action potential, the membrane is completely refractory (unexcitable) to further stimulation.
The cell is refractory because most of its Na+ voltage-gated channels are inactivated and cannot be reopened until the membrane is repolarized.
This means that no matter how strongly the cell is stimulated, it is unable to fire a second action potential
this unresponsive state is called the absolute refractory period.
When a cell is partly depolarized, the pool of excitable voltage-gated Na+ channels is reduced
consequently, a stimulus may not be able to recruit a sufficient number of Na+ channels to generate an action potential
A chemical explosion requires a critical mass of material
the spike of the action potential can be generated only if a critical number of Na+ channels are recruited (activated),
this is the threshold value.
THE STATES OF Na+ AND K+ CHANNELS CORRELATEDWITH THE COURSE OF AN ACTION POTENTIAL
Insert THE STATES OF Na AND K CHANNELS CORRELATED WITH THE COURSE OF AN ACTION POTENTIAL
The action potential can be viewed in terms of the flow of charged ions through selective ion channels as illustrated above:
figure “D” shows that during the after-hyperpolarization , the sodium channels are closed and the potassium channels remain in their active state
note the K+ channels have no inactivated state
the K+ channels will close and the Na+ channels will become excitable with the membrane potential returning to its resting level
the potassium channels open more slowly and, therefore, have not yet responded to the depolarization
figure “B” shows that during the depolarization phase of the action potential, the voltage-gated Na+ channels are activated (open)
the potassium channels open more slowly and, therefore, have not yet responded to the depolarization
opening of the activation gate allows sodium ions to flow into the cell which results in depolarization
. figure “A” shows the resting membrane potential with both Na+ and K+ voltage-gated ion channels closed (inactive but excitable)
the cell membrane is in a “resting state” with no ion exchange through the voltage--gated channels
However, before the membrane potential reaches ENa, sodium permeability begins to decrease and potassium permeability increases
This change in membrane conductance again drives the membrane potential towards EK, accounting for repolarization of the membrane.
During the rising phase, the cell membrane becomes more permeable to sodium;
as a consequence, the membrane potential begins to shift more towards the equilibrium potential for sodium
As discussed earlier, the depolarizing and repolarizing phases of the action potential can be explained by the relative changes in membrane conductance (permeability) to sodium and potassium ions
THE GENERATION OF AN ACTION POTENTIAL
Insert the Generation of An action potential
Before and after the action potential (“1” and “9” in upper figure), the cell membrane is at its resting membrane potential of -70 mV.
The lower graph shows the action potential can be divided into three phases:
the after-hyperpolarization phase
potassium continues to leave the cell through both voltage-gated and K+ leak channels, and the cell hyperpolarizes (“7” in upper figure)
the voltage-gated K+ channels have not yet closed
occurs because when the falling membrane potential reaches -70 mV
the falling phase (repolarization)
corresponds to an increase in K+ permeability
as K+ moves out of the cell, the membrane potential rapidly becomes more negative, creating a falling phase of the action potential (“6” in upper figure) and moving the cell towards its resting potential
at a positive membrane potential, the concentration and electrical gradients for K+ favors movement of K+ out of the cell
the rising phase (depolarization)
due to a sudden temporary increase in the cell’s permeability to Na+, causing the inside of the cell to progressively become less negative and possibly positive (> 0 mV)
an action potential begins when a graded potential reaching the trigger zone (“2” in upper figure) depolarizes the membrane to threshold (-55 mV, “3” in upper figure)
The figure above illustrates the voltage and ion permeability changes that take place in one section of the membrane during the generation of an action potential.
Action potentials occur when voltage-gated ion channels open, altering membrane permeability to Na+ and K+.
PHASES OF AN ACTION POTENTIAL
Insert PHASES OF AN ACTION POTENTIAL
The action potential is a transient change in the membrane potential from its resting level (the resting membrane potential) characterized by
afterhyperpolarization
regulated membrane ion exchange (Na+/K+ pump) will return the membrane potential to its normal resting level
the efflux of positive ions may continue long enough for the membrane potential to become a more negative value than its resting membranepotential
repolarization
the efflux of positive ions out of the cell will result in the membrane potential returning back towards its resting level
an overshoot
the influx of positive ions may continue long enough for the
membrane potential to become a positive value
a rapid rising phase - a significant influx of positive ions becomes great enough for the membrane potential to become less negative (depolarize)
this increase in the membrane potential will initiate the mechanisms responsible for the propagation of the action potential
a gradual depolarization from the resting level to threshold
the membrane potential becomes more positive due to an influx of positive ions
action potentials depend on a regenerative wave of ion channels opening and closing
at point “C”, the stimulus is even stronger and the local potential change barely reaches the level to elicit an action potential, called the threshold
at point “B”, the stimulus is greater but the intensity is still not enough to excite the cell membrane
the stimulus is strong enough to disturb the membrane potentially locally
these local potential changes are called acute local potentials and when they fail to elicit and action potential, they are called acute subthreshold potentials
at point “A” a weak stimulus causes the membrane potential to change from -90 mV to -85 mV, but this change is not sufficient for the automatic regenerative processes of the action potential to develop
a very weak stimulus may not be able to produce any change in the resting membrane potential
Basically, any factor that causes sodium ions to begin to diffuse inward through the membrane in sufficient numbers will set off the automatic regenerative opening of the sodium ion channels.
This can result from
passage of electricity through the membrane
chemical effects on the membrane
simple mechanical disturbance of the membrane
TERMINOLOGY ASSOCIATED WITH CHANGESIN THE MEMBRANE POTENTIAL
Insert TERMINOLOGY ASSOCIATED WITH CHANGESIN THE MEMBRANE POTENTIAL
If the cell’s permeability to an ion changes, the change in the cell’s membrane potential can be monitored by recording electrodes (illustrated in the above figure).
When the resting membrane potential moves away from the 0 mV becoming more negative, it is called hyperpolarization.
A return to the resting membrane potential is termed repolarization.
If the membrane potential difference is increasing, the value of Vm must be moving away from the ground value of 0 mV and becoming more negative.
When the trace moves upward (becomes less negative), the potential difference between the inside of the cell and the outside (set at 0 mV) decreases, and the cell is said to have depolarized
If the membrane potential difference is decreasing, the value of Vm is moving closer to the ground value of 0 mV and is becoming less negative.
The membrane potential (Vm) begins at a steady resting value of -70 mV
Conditions Leading to Arrhythmias
There are three different conditions that have the capability of causing an impulse to
continue to travel around the circle, one or more of these conditions must exists along
with a unidirectional block to allow myocytes to be stimulated during the relative
refractory phase and initiate re-entry → arrhythmias
a shortening of the absolute refractory period
can be caused by repeated electrical stimulation and epinephrine
administration during cardiac resuscitation (!!!)
a decrease in action potential impulse conduction velocity
examples: ischemia and hyperkalemia
- both conditions will result in a more positive resting membrane potential,
therefore increasing the number of non-excitable Na ion channels →
decreases slope and magnitude of phase 0 = decrease action potential
conduction velocity → increased potential for developing an arrhythmia
an increase in action potential impulse pathway length, therefore it takes a longer time to travel the circuit (shown in figure above)
examples: hypertrophy and cardiomyopathy
Effects of Extracellular Potassium
In the clinical scenario, the concentration of K+ in the blood and interstitial space is the major determinant of the resting membrane potential.
If K+ concentration in the blood moves out of the normal range of 3.5 - 5.0 mmol/L, the result is a change in the resting membrane potential of cells.
Clinically it is extremely important to understand how the extracellular K+ level influences the resting membrane potential; it can effect both neural and muscle (skeletal, smooth, and cardiac) activity.
Primary Hyperkalemic Paralysis and Effects on Na+ Channels
In an inherited disorder called primary hyperkalemic paralysis, patients have episodes of painful spontaneous muscle contractions (“muscle spasms”) followed by periods of paralysis of the affected skeletal muscle. These symptoms are accompanied by and caused by increased blood plasma and interstitial fluid potassium ion concentrations.
As the depolarization of cells increases, the cells become refractory because of inactivation of the voltage-gated Na+ channels (the inactivation gates are closed due to the less negative membrane potential).
Consequently, the cells become unable to fire action potentials and are not able to contract in response to action potentials generated within their motor axons (paralysis).
A pathological condition which is due to an abnormal “leakage” of intracellular potassium ions will result in an elevation in extracellular [K+] causing the resting membrane potential of the skeletal muscle cells to become more positive.
Some patients with this disorder have mutations of voltage-gated Na+ channels that result in a decreased rate of voltage inactivation, which results in longer-lasting action potentials in skeletal muscle cells and increased efflux of K+ during each action potential.
This will result in an increase in the extracellular [K+].
if the blood level falls too low, hyperkalemia, the resting membrane potential becomes more negative (hyperpolarized) moving farther from threshold
a stimulus of normal strength will not reach the threshold value (figure d)
if the blood K+ level is abnormally elevated, hyperkalemia (figure c), the membrane potential becomes more positive and shifts the resting membrane potential closer to threshold
this causes the cells to fire action potentials in response to smaller graded potentials
at normal K+ levels, subthreshold graded potentials (figure a) do not trigger action potentials, and suprathreshold graded potentials do (figure b)
Resting Membrane Potential
Cells have an electrical potential difference (voltage difference), called the resting membrane potential, across their plasma membranes.
The cytoplasm is usually electrically negative relative to the extracellular fluid.
The resting membrane potential is necessary for the electrical excitability of neurons, skeletal muscle, smooth muscle, and the heart.
The difference in potential energy of an ion across a membrane depends on its concentration difference and the electrical potential difference across the membrane.
Variability in Resting Membrane Potentials across different Tissues
The resting membrane potential is not stable in all cells of the body and the variability is associated with the tissue’s function.
Changes in membrane potential are associated with mechanical, electrical, and chemical stimulation plus spontaneous depolarization.
SA Nodal Cells
The initial potential is only -50 to -70 mV because the nodal cells possess fewer K+ leak channels, which are responsible for the big, stable resting membrane potential of atrial and ventricular myocytes.
fewer active ion channels → fewer positive ions leaving the cell → more positive ions are left within the cell → resting membrane potential becomes more positive (or remains positive).
The membrane potential for the SA node is small and unstable (figure on the right).
Smooth Muscle Cells
The smooth muscle of the gastrointestinal tract is subject to almost continual but slow electrical activity (figure on the left). This activity tends to have two basic types of electrical waves:
Spike/Pacemaker Potentials
. spike potentials are true action potentials which occur automatically when theresting membrane potential (RMP) becomes more positive than about -40 to -45 mV
(note: the normal resting membrane potential is between -50 and -60 mV)
Slow Wave Potentials
occur rhythmically and are slow undulating changes in the resting membrane potential,
thought to be due to a slow undulation of the Na+/K+ pump activity
Measuring Membrane Potential Difference
Reading the Measurement
Equipment
CHart Recorder
A chart recorder connected to the voltmeter can make a recording of the membrane potential versus time.
Reference Electrode
A reference electrode is placed in the external bath, which represents the extracellular fluid.
In living systems, by convention, the ECF is designated as the ground and assigned a charge of 0 mV.
REcording Electrode
. A recording electrode is inserted through the cell membrane into the cytoplasm of the cell.
When the recording electrode is placed inside a living cell, the voltmeter measures the membrane potential;the electrical difference between the ICF and the ECF.
Voltemeter
Micropipettes are then connected to a voltmeter, which measures the electrical difference between two points in units of either volts (V) or millivolts (mV).
When the recording electrode is placed inside a living cell, the voltmeter measures the membrane potential;the electrical difference between the ICF and the ECF.
Electrodes/Micropipettes
Electrodes are created from hollow glass tubes drawn to very fine points. These micropipettes are filled with a fluid that conducts electricity
Conditions Required for Actional Potential in the Refractory Period
Conditions Required for Resting Membrane Potential
Balance of Ion Exchange
Na+/K+ - ATPase Pump
The most abundant ion pump in higher organisms is the sodium-potassium ion pump, also known as Na+/K+-ATPase. It is found in the plasma membrane of practically every eukaryotic cell and is responsible for maintaining the low sodium and high potassium concentration in the cytoplasm. The sodium-potassium pump is an integral membrane protein consisting of two subunits, one large and one small. Sodium ions are transported out of the cell and potassium ions are brought into the cell.
Comparison of Resting Membrane Potential with Pump Added to Membrane
the resting membrane potential generated when the membrane is permeable to both sodium ions (+61 mV) and potassium ions (-94 mV) plus pumping of both of these ions by the Na+/ K+ -ATPase pump = -90 mV
the resting membrane potential generated when the membrane is permeable to both sodium ions (+61 mV) and potassium ions (-94 mV) = -86 mV
this occurs when the [K+]i = 140 mEq/L and [K+]o = 4 mEq/L
- the ratio is: [K+]i / [K+]o = 35.0
this occurs when the [Na+]i = 14 mEq/L and [Na+]o = 142 mEq/L
- the ratio is: [Na+]i / [Na+]o = 0.1
the resting membrane potential generated when the membrane is onlypermeable to potassium ions = -94 mV
this occurs when the [K+]i = 140 mEq/L and [K+]o = 4 mEq/L
Mechanisms
During one cycle, three Na+ are exchanged for two K+, and one ATP molecule is hydrolyzed to ADP and phosphate (Pi), illustrated in steps 4 - 5 above.
The sodium-potassium pump is said to be an “electrogenic pump” because it creates an electrical potential across the cell membrane as it cycles, adding to its influence on the resting membrane potential.
The fact that the number of ions exchanged is not equal, a net movement of positive ions out of the cell occurs creating positivity outside the cell and leaves a deficit of positive ions inside the cell; that is, it causes negativity on the inside.
Subsequent binding of extracellular K+ and dephosphorylation return the protein to its original form and transfer K+ into the cell.
Binding of intracellular Na+ and phosphorylation by adenosine triphosphate (ATP) inside the cell may induce a conformational change that transfers Na+ to the outside of the cell (steps 1 - 3).
It also plays a role in the resting membrane potential by maintaining ion gradients which are required for the ion “leak” channels to function.
For a cell to have a steady resting membrane potential, the charge separation across the membrane must be constant; the influx of positive charge must be balanced by the efflux of positive charge. If these fluxes were not equal, the charge separation across the membrane, and thus the membrane potential, would vary continually. Therefore, for the cell to achieve a resting state, the movement of K+ out of the cell must balance the movement of Na+ into the cell. Although these steady ion leaks cancel each other, they can not be allowed to continue unopposed for any appreciable length of time. Otherwise, the [K+]i would be depleted and the [Na+]i would increase, and the ionic gradients would gradually run down, reducing the resting membrane potential.
Dissipation of ionic gradients is prevented by the Na+/K+ -ATPase pump, which extrudes 3Na+ from the cell while taking in 2K+. Because the pump moves these two ions against their net electrochemical gradients, energy must be provided to drive these actively transported fluxes. The energy comes from the hydrolysis of ATP.
It maintains a high intracellular potassium concentration, which is necessary for protein synthesis.
The pump counterbalances the tendency of sodium ions to enter the cell passively and the tendency of potassium ions to leave passively.
Diffusion Potentials (no active transport)
Golfman-Hodgkins-Katz Equation
The importance and meaning of the Goldman-Hodgkin-Katz equation are
the permeabilities of the sodium and potassium channels undergo rapid changes during conduction of an action potential
a positive ion concentration gradient from inside the membrane to the outside causes electronegativity inside the membrane
the degree of importance of each of the ions in determining the voltage is proportional to the membrane permeability for that particular ion
the permeability of the membrane to potassium is about 100 times greater as to sodium
- using this value in the Goldman equation gives an internal membrane potential of -86 mV, which is near to the potassium potential shown above
the resting membrane potential will be primarily determined by the most permeable ion and the voltage will be closest to the electrical potential of that ion
sodium, potassium, and chloride ions are the ions most importantly involved in the
development of the membrane potential
Thus the following formula, called the Goldman-Hodgkin-Katz equation, gives the calculated membrane potential on the inside of the membrane when the membrane is permeable to more than one univalent ion:
EMF (mV) = log CNa+i PNa+ + CK+i PK+ + CCl-o PCl-
CNa+o PNa+ + CK+o PK+ + CCl-i PCl-
Three factors affecting diffusion potentials
the concentrations (C) of the respective ions on the inside (i) and outside (o)
the permeability of the membrane (P) to each ion
the polarity of the electrical charge of each ion
Electrical potentials exist across the membranes of essentially all cells of the body. The above figure illustrates the generation of the equilibrium potential for both potassium and sodium ions, there is no active transport.
note that the internal membrane potential is negative when potassium ions diffuse out and positive when sodium ions diffuse in because of the opposite concentration gradients of these two ions
the establishment of an equilibrium potential when the membrane is permeable to only to sodium ions is shown in figure B
• the equilibrium potential for sodium is +61 mV, this is the voltage required at the inside surface of the membrane for the net diffusion of sodium to be zero; the electrical gradient force is equal but opposite to the chemical gradient force
. the establishment of an equilibrium potential across a cell membrane, caused by potassium ions diffusing from inside the cell to the outside through a membrane selectively permeable only to potassium (shown in figure A)
the equilibrium potential for potassium is -94 mV, this is the voltage required at the inside surface of the membrane for the net diffusion of potassium tobe zero. - the electrical gradient force is equal but opposite to the chemical gradient force
Ion Potentials
A membrane’s selectivity for permeant ions is determined by the relative proportions of various types of ion channels.
Normal Ion Concentraions
Calcium
Intracellular = 0.0001 nM
Extracellular = 1 mM
Chloride
Eion at 37degrees = -63mV
Intracellular = 10 nM (5-15)
Extracellular = 108nM (bw 100-108nM)
Eion at 37degrees = +60mV
Intracellular = 15nM
Extracellular = 145nM (135-145)
Eion at 37degrees = -90mV
Intracellular = 150 nM
Extracellular = 5nM (bw 3-5nM)
Sodium
when the Na+ concentration is 150 mM outside and 15 mM inside, the equilibriumpotential for Na+ is about +60 mV
the concentration gradient moving Na+ into the cell is exactly opposed by a positive membrane potential of +60 mV
meanwhile, Cl- left behind in the extracellular fluid gives that compartment a negative charge
this imbalance creates an electrical gradient that tends to “pull” Na+ back out of the cell
Because Na+ is more concentrated outside the cell, some Na+ moves into the cell and accumulates there.
The same artificial cell model used in the previous page (K+, Pr-, and Na+ inside; Na+ and Cl- outside) is now used to explain the effects of making the membrane only permeable to Na+.
Potassium
Excitable cells have nongated channels that for the most part are selectively permeable to K+ ions when the cell is at rest
The flux of K+ across the membrane is determined by both the K+ concentration gradient and the electrical potential across the membrane:
Calculating the Electrochemical Equilibrium for Potassium Ions
EA - EB = -60 mV x log [K+]A
[K+]B
Note that an electrical potential difference of about 60 mV is required to balance a 10-fold concentration difference of a univalent ion (this is a useful rule of thumb to remember).
The Nernst equation reveals that at an electrical potential difference (EA - EB) of -60 mV, K+ is in electrochemical equilibrium; K+ tends to move from side B to side A due to the electrical potential difference, which counteracts the tendency for K+ to move from side A to side B due to the concentration difference.
Effects of Inserting Potassium Leak Channels into a theoretical cell
A potassium ion leak channel is inserted into the cell membrane and potassium leaks out of the cell because of the concentration gradient (figure b):
When the electrical force attracting K+ into the cell becomes equal in magnitude to the chemical concentration gradient driving K+ out of the cell, there is an equilibrium potential generated.
Because opposite charges attract each other, the negative proteins inside the cell try to pull K+ back into the cell (figure c).
the loss of positive ions from the cell creates an electrical gradient
if only the force acting on K+ were the concentration gradient, K+ would leak out until he K+ concentration inside the cell equaled the K+ concentration outside
as K+ leaves the cell, the negatively charged proteins (Pr-) are unable to follow which results in a gradual build up of a negative charge on the inside of the cell
However, it is not in chemical equilibrium:
all the ions would all diffuse down their respective concentration gradients if they could cross the cell membrane
there are concentration gradients for all four types of ions in the system
The cell is placed in a solution of Na+ and Cl-. Both the cell and the solution are electrically neutral, and the system is in electrical equilibrium.
The artificial cell in figure (a) has a membrane that is impermeable to ions and large negatively charged proteins, represented by Pr-.
the continued efflux of K+ builds up an excess of positive charge on the outside of the cell and leaves behind on the inside an excess of negative charge (figure B)
this buildup of charge acts to impede the further efflux of K+, so that eventually an equilibrium is reached
when the electrical and chemical driving forces are equal and opposite, this is known as the equilibrium potential
. in a cell permeable only to K+ the resting membrane potential is generated by the efflux of K+ down its concentration gradient (figure A)
Electrochemical Potential Difference
A membrane separates aqueous solutions in two chambers, A and B, in this figure:
1. ion X+ is at a higher concentration on side A than on side B
2. if no electrical potential differences exists between A and B, a concentration force for
X+ tends to cause X+ to flow from A to B (yellow arrow)
• just as if it were an uncharged particle
3. if side A is electrically negative with respect to B, an electrical force tends to cause
X+ to flow from B to A (red arrow)
4. the direction of net X+ movement depends on whether the effect of the concentration
difference or the effect of the electrical potential difference is larger
• by comparing the two tendencies, the direction of net X+ movement can be predicted
The quality that allows a comparison of the relative contributions of ionic concentration differences and electrical potential differences across a membrane is called the electrochemical potential difference (Δµ) of an ion. The electrochemical potential difference of X+ across the membrane is defined as:
Δµ(X) = µA (X) - µB (X) = RT ln[X]A/[X]B + zF(EA - EB)
The different passive and active transport systems are coordinated in a living cell to maintain intracellular ions and other solutes at a concentration compatible with life.
Consequently, the cell does not equilibrate with the extracellular fluid but exists in a steady state with the extracellular solution.
Maintenance of this steady state with ion concentrations inside the cell different from those outside the cell is the basis for the difference in electrical potential across the plasma membrane.
the gradient of electrochemical potential across the membrane for a specific solute is based on the sum of 2 forces: the electric potential force and the chemical potential force.
. Net movement of an ion into or out of a cell continues as long as the driving force exists.
Net movement stops and equilibrium is reached only when the driving force of electrochemical potential across the membrane becomes zero.
Nernst Potential
The potential level across the membrane that prevents net diffusion of an ion in either direction through the membrane is called the Nernst potential for that ion.
Nernest Equation
EMF = +/- 61 xlog (conc inside)/(Conc outside)
The magnitude of this potential is determined by the ratio of the ion concentration on the two sides of the membrane - the greater this ratio, the greater the tendency for the ions to diffuse in one direction.
The above equation can be used to calculate the Nernst potential for any univalent ion, usually assuming that
-the potential outside the membrane always remains at exactly zero potential,
- and the Nernst potential that is calculated is the potential inside the cell
Chemical Potential Difference
the force resulting from the difference in the concentration of a substance between the
inside and outside of the cell
• all substances tend to move from areas of high concentration to areas of low
concentration
Electrical Potential Difference
the difference in electrical potential between the two sides of the membrane and
applies only to ions and other charged solutes
• positive ions tend to move toward the negative side, whereas negative ions tend to
move toward the positive side
Separation of CHarges
In the body, separation of charge takes place across the cell membrane.
An active transport carrier is now inserted into the membrane and uses energy to move positive ions out of the cell against their concentration gradient shown in figure (b).
When positive ions are pumped out of the cell this creates a separation of charge known as a dipole layer.
When positive ions are pumped out of the cell, they line up along the outside of the membrane, and on the inside, the negative ions that have been left behind line up. This creates a separation of electrical charge across the cell membrane consisting of a thin cloud of positive and negative ions spread over the inner and outer surfaces of the membrane, this is known as a dipole layer.
Creation of the Electric Capacitator
This arrangement of ions on either side of the lipid bilayer results in the membrane acting as an “electrical capacitor”; only minimal amount of positive ions must be transported outward to create an electrical dipole layer
less energy is required to reestablish normal ion concentrations when disrupted
this allows for very rapid changes in the membrane potential
an incredibly small number of ions need to be transferred through the membrane to establish a membrane potential of -90 mV
• only 1/5,000,000 to 1/100,000,000 of total positive charges inside the cell
. the charge separation gives rise to an electrical potential difference across the membrane - The resting membrane potential
Principle of LEctric Neutrality
everywhere except adjacent to the surfaces of the cell membrane itself, the negative and positive charges in the cytoplasm are equal
- this is called the principle of electrical neutrality
- it is directly proportional to the charge separation across the membrane
this separation of charge is maintained because the lipid bilayer acts as a barrier to the diffusion of ions
at rest, the cell has an excess of positive charges on the outside of the membrane and an excess of negative charges on the inside
the excess of both charges on either side of the membrane at rest represents a small
fraction of the total number of ions inside and outside the cell membrane
In living systems, electrical gradients are measured on a relative scale rather than an absolute scale because we cannot measure electrical charge as numbers of electrons gained or lost.
Physiological measurements are always on a relative scale, on which the ECF is assigned a value of zero. This shifts the scale to the left and gives the inside of the cell a relative charge of -2 (figure c).
. On an absolute charge scale, the extracellular fluid (ECF) would be at +1 and the intracellular fluid (ICF) would be at -1.
the input of energy to transport ions across the membrane has created an electrical gradient, a difference in the net charge between two regions
. as soon as the first positive ion leaves the cell, the electrical equilibrium between the ECF and ICF is disrupted
• the cell’s interior has a net charge of -1 while the cell’s exterior has a net charge of +1 (absolute charge)
The phospholipid bilayer of the artificial cell, like the bilayer of a real cell, acts as an insulator to prevent free movement of ions between the intracellular and extracellular compartments.
This makes the system shown in figure (a) to be in osmotic, chemical, and electrical equilibrium.
Water can freely cross this cell membrane, making the extracellular and intracellular osmotic pressures equal.
The cell can be placed in a solution that is also electrically neutral and contains the same types of positive and negative ions
. The cell is placed in an aqueous solution, also electrically neutral, that contains the same types of cations and anions.
In theory, a cell can be filled with neutral molecules that dissociate into positive and negative ions
The diagram in figure (a) illustrates an artificial cell filled with molecules that dissociate into positive and negative ions, represented by the plus and minus signs. Because the molecules were electrically neutral to begin with, there are equal numbers of positive and negative ions inside the cell.
Structural Support
Communication between the Cell and Its Environment
Regulation of Exchange with Environment
classifications
There are two ways to categorize the energy requirements required to move molecules across cell membranes:
energy requirements
insert MAP OF ENERGY REQUIREMENTSFOR MEMBRANE TRANSPORT
Classification
active transport
requires the input of energy from some outside source, such
as the high-energy phosphate bond of ATP
passive transport
THE PASSIVE MOVEMENT OF SOLUTES ACROSS A CELL MEMBRANE
THE RATE OF SOLUTE TRANSPORT BY SIMPLE DIFFUSION
In most physiological systems, membrane thickness is a constant (stable); therefore Fick’s law equation can be rearranged to:
Diffusion rate = concentration gradient x membrane permeability
Surface area
The rules for simple diffusion can be combined mathematically into an equation known as Fick’s law of diffusion, a relationship that is expressed as:
Insert Fick's Law of DIffusion
Rate of diffusion = surface area x concentration gradient x membrane permeability
membrane thickness
Membrane permeability is the most complex (variable) of the four terms in Fick’s law because several factors influence it:
the composition of the lipid bilayer across which it is diffusing
the lipid-solubility of the molecule
the size of the diffusing molecules
The rate of solute entry increases linearly with extracellular concentration, increasing the extracellular concentration increases the gradient that drives solute entry.
The graph on the right shows solute transport across a plasma membrane by simple diffusion according to concentration gradients; inside vs. outside, high to low.
DIFFUSION THROUGH A LIPID BILAYER
insert DIFFUSION THROUGH A LIPID BILAYER
Diffusion across a membrane has no preferential direction; it can occur from the outside of the cell toward the inside or visa versa.
This results in a net movement of solute from left to right across the membrane until the concentration of solute is the same on both sides (figure on the left).
Initially, random movement from left to right across the membrane is more frequent than movement in the opposite direction because there are more molecules on the left.
the solute molecules move randomly by Brownian movement.
The diffusion of a solute across a cell membrane is driven by the difference in the concentration on the two sides of the membrane;
Two types
facilitated diffusion
simple diffusion
An example of simple diffusion
OSMOSIS AND OSMOTIC PRESSURE
THE MEASUREMENT OF OSMOTIC PRESSURE
insert THE MEASUREMENT OF OSMOTIC PRESSURE
TONICITY DEPENDS ON THE RELATIVE CONCENTRATION OF NONPENTRATING SOLUTES
Insert TONICITY DEPENDS ON THE RELATIVE CONCENTRATION OF NONPENTRATING SOLUTES
ECF AND ICF CHANGES WHEN AN ISOTONIC AND HYPERTONIC SOLUTION ARE ADDED
Body fluid volumes and plasma osmolality are kept constant by three major aspects to control water balance:
drinking habits and the perception of thirst
excretion of water by the kidneys
arginine vasopressin (antidiuretic hormone, ADH
People normally stay in a stable water balance
that is, water input and output are equa
The effects of infusing intravenously 1.0 L of a 5% NaCl solution (osmolality about 1580 mOsm/Kg H2O) are shown in figure “D”:
the addition of a hypertonic saline solution to the ECF increased the volume in the extracellular compartment in part because of water loss from the intracellular compartment
the final ECF volume is increased by 3.7 L
2.7 L from the intracellular comparment
1 L from the intravenous infusion
a new equilibrium will be established, with a final osmolality higher than normal (about 315 mOsm/Kg H2O) but equal inside and outside of the cells
all the salt stays in the ECF which creates a hypertonic environment for the cells
water leaves the cells leaving behind the solutes making the ICF more concentrated
Since isotonic saline is isosmotic to plasma or ECF by definition, when 2.0 L of isotonic saline (0.9% NaCl solution) is added to the ECF (figure C), there is no change in cell volume
Therefore all of the isotonic saline is retained in the ECF and there is no change in osmolality.
ECF AND ICF CHANGES WHEN PURE WATER IS ADDED
When 2 L of pure water is added to the ECF (figure B)
the new total body water volume is 44 L
⅔ of the added water ends up in the intracellular compartment and ⅓ stays in the ECF
no solute was added, so the new osmolality at equilibrium is 272 mOsm/Kg H2O
plasma osmolality is lowered and water moves into the cell compartment along the osmotic gradient
the intracellular osmolality falls until a new equilibrium (solid lines) is achieved
the entry of water into the cells causes them to swell (water is therefore hypotonic)
In the normal situation (figure A)
the osmolality of both fluids is 285 mOsm/Kg H2O.
⅓ in the ECF (14 L)
⅔ of total body water (28 L) is in the ICF
In the above figures
the dashed lines indicate the normal condition, and the solid lines the situation after a new osmotic equilibrium has been attained
the area of a box gives the amount of solute present in a compartment
note that the height of the boxes is always equal, because osmotic equilibrium (or equal osmolalities) has been achieved
the x-axis the volume (L)
the y-axis represents total solute concentration or osmolality (mOsm/Kg H2O)
The distribution of water between intracellular and extracellular compartments changes in a variety of circumstances.
If the osmolality changes in one compartment, water moves so as to restore a new osmotic equilibrium
ICF and ECF are in osmotic equilibrium because of the high water permeability of cell membranes, which does not permit an osmolality difference to be sustained
Despite the different compositions of ICF and ECF, the total solute concentration (osmolality) of these two fluid compartments is normally the same
Tonicity
The differences between tonicity and osmolarity are
osmolarity alone does not predict what happens to a cell when placed in a solution
tonicity by definition predicts what happens to cell volume when the cell is placed in the solution
osmolarity can be used to compare two solutions, and the relationship is reciprocal (solution A is hyperosmotic to solution B; therefore , solution B is hyposmotic to solution A);
tonicity always compares an extracellular solution to the intracellular fluid
osmolarity describes the number of particles dissolved in a volume of solution and it has units (ex. osmoles/liter);
tonicity has no units and is only a comparative term
key in predicting tonicity is knowing the relative concentrations of nonpenetrating solutes in the cell and in the solution:
if the concentrations of nonpenetrating solutes are the same in the cell and the solution, there is no net movement of water at equilibrium
the solution is isotonic to the cell, no change in intracellular volume (figure b)
if the cell has a lower concentration of nonpenetrating solutes than the solution, there will be net movement of water out of the cell
the cell shrinks and the solution is hypertonic
if the cell has a higher concentration of nonpenetrating solutes than the solution, there will be net movement of water into the cell
the cell swells and the solution is hypotonic (figure a)
; it describes the cell volume once the cell has come to equilibrium with the solution
the physiological term used to describe a solution and how that solution affects cell volume
The terms osmolarity and osmolality are frequently confused and incorrectly interchanged:
osmolality
number of molecules dissolved in 1 kg of solvent (water)
usually used in clinical situations because it is easy to estimate people’s body water content by weighing them
expressed as osmoles of solute/Kg H2O
independent of temperature
Example 2
add NaCl which dissociates into two ions
thus 1 mole NaCl results in 2 moles of particles
1 M NaCl x 2 ions per NaCl = 2 Osm NaCl
one of Na and one of Cl = a 2 Osm NaCl
example 1
one mole of glucose dissolved in enough water to create 1 liter of solution yields a 1 molar solution (1 M),
1 M glucose x 1 particle per glucose molecule = 1 OsM glucose
glucose does not dissociate therefore
osmolarity = concentration x number of dissociable particles
mOsm/L = mmol/L x number of particles/mol
measurements of osmolarity are dependent on temperature because the volume of solvent varies with temperature
refers to the osmotic pressure generated by the dissolved solute molecules in 1 L of solvent
DISTRIBUTION OF SOLUTES IN THE BODY FLUID COMPARTMENTS
Insert DISTRIBUTION OF SOLUTES IN THE BODY FLUID COMPARTMENTS
The major difference between interstitial fluid and plasma is that the plasma contains significantly more protein (primarily albumen).
The composition of the plasma (P) and interstitial fluid compartments (I) of the ECF is similar because they are separated only by the capillary endothelium
, a barrier that is freely permeable to ions and small molecules.
Principle among these transporters is the Na+/K+-ATPase, which converts the energy in ATP into ion and electrical gradients, which in turn can be used to drive the transport of other ions and molecules.
The composition of the of the intracellular fluid (C ) is maintained by the action of various specific membrane transport proteins.
The distribution of solutes between the extracellular and intracellular fluid is summarized in the above set of graphs.
Osmotic Pressure
units of the osmotic pressure are mm Hg
the pressure necessary to stop net movement of water across a selectively permeable membrane that separates the solution from pure water.
the pressure that must be exerted to exactly oppose the osmotic movement off ater into compartment B is the osmotic pressure of solution B
water can move across the membrane freely by osmosis, thus water moves from compartment A to dilute the more concentrated solution in compartment B
a concentration gradient across the membrane exist for glucose, but the membrane is not permeable to glucose
The driving force for the movement of water across the plasma membrane is the difference in water concentration between the two sides of the membrane.
OSMOSIS ACROSS A SEMIPERMEABLE MEMBRANE
insert OSMOSIS ACROSS A SEMIPERMEABLE MEMBRANE
Osmosis
Water is able to move freely between cells and the extracellular fluid, and will distribute itself until water concentrations are equal throughout the body
the body is in a state of osmotic equilibrium.
Osmosis is a passive transport mechanism that tends to equalize the total solute concentrations of the solutions on both sides of every membrane.
Osmosis can be viewed as the movement of water from a solution of high water concentration (low concentration of solute) toward a solution with a lower concentration of water (high solute concentration).
a solution with a high concentration of solutes has a low concentration of water and visa versa.
The spontaneous movement of water across a membrane driven by a gradient of water concentration is the process known as osmosis.
Concentration
the number of particles per unit volume:
Diffusion has the following seven properties:
diffusion can take place in an open system or across a partition that separates two systems
diffusion is inversely related to molecular size;
the bigger → the slower
diffusion is directly related to temperature
diffusion is rapid over short distances but much slower over long distances
net movement of molecules continues until the concentration is equal everywhere
molecules move from an area of high concentration to an area of lower concentration
diffusion is a passive process which means the energy used for molecular movement is only the kinetic energy possessed by all molecules
The net result of diffusion is the movement of substances according to their difference in concentrations, from regions of high concentration to regions of low concentration
This movement, known as diffusion, is a result of the spontaneous Brownian (random) movement that all molecules experience.
Any solute will tend to uniformly occupy the entire space available to it.
does not require the direct input of energy
physical requirements
transported by using vesicles
transported with the aid of a membrane protein
for movement through the phospholipid bilayer
Mediated Transport
Active Transport
for larger molecules
pinocytosis
phagocytosis
endocytosis
Secondary
The sodium ion concentration gradient, with Na+ concentration high in the extracellular fluid and low in the intracellular fluid, is the source of potential energy that the cell can harness for other functions. Membrane transporters that use potential energy stored in concentration gradients to move molecules are called secondary active transport systems. Secondary active transport uses the kinetic energy of one molecule moving down its concentration gradient to push other molecules against their concentration gradient. As sodium ions move into the cell, it either brings one or more molecules with it or trades places with molecules existing in the cell. The most common secondary active transport systems dependent on Na+ are listed on the left in the above figure. Note that the cotransport substances may be either other ions or uncharged molecules, such as glucose.
Examples
Absorption of AAs
The primary products of protein digestion are free amino acids, dipeptides and tripeptides, all of which can be absorbed. Amino acid structure is so variable that multiple amino acid transport systems are found in the intestine; most free amino acids are carried by Na+-dependent cotransport proteins. A few amino acids transporters are H+ dependent.
Dipeptides and tripeptides are carried into the mucosal cell using H+-dependent cotransport. Once inside the epithelial cell, these peptides have two possible fates:
1. most are digested by cytoplasmic peptidases into amino acids, which are then
transported across the basolateral membrane and into the circulation
2. those that are not broken down to amino acids are transported intact across the
basolateral membrane
♥ the transport system that moves these peptides also is responsible for
intestinal uptake of certain drugs
- beta-lactam antibiotics
- angiotensin-converting enzyme (ACE) inhibitors
- thrombin inhibitors
Secondary Active Transport in the GI Tract
Intestinal glucose and galactose absorption uses transporters identical to those found in the renal proximal tubule: an apical Na+-glucose SGLT symporter (SGLT1) and a basolateral GLUT2 transporter, these transporters move galactose as well as glucose. Fructose absorption, however, is not Na+-dependent. Fructose moves across the apical membrane by facilitated diffusion on the GLUT5 transporter and across the basolateral membrane by GLUT2.
non sodium dependent transporters
Sodium Dependent Carriers
Primary
H+k+ - ATPase
H+ - ATPase or proton pump
ca2+ - ATPase
Na+/k+ - ATPase
Facilitated Diffusion
For many solutes of physiological importance, such as sugars and amino acids, the relationship between transport rate and concentration difference follows a curve that reaches a plateau (figure on the right). Furthermore, the rate of transport of these hydrophilic substances across the cell membrane is much faster than expected for simple diffusion through a lipid bilayer, this was found to be due to facilitated diffusion.
Facilitated diffusion refers to the process of using protein carrier-mediated transport because it uses an integral membrane protein, the carrier (usually very specific), that binds to the transported solute on one side of the membrane and releases it at the other side. No outside source of energy is needed to move molecules across the cell membrane. Facilitated diffusion only mediates the transport of a substance down its chemical or electrochemical gradient, they can not move substances against concentration gradients. Facilitated diffusion acts to equalize concentrations of transported substances on the two sides of the membrane, and net movement stops at equilibrium.
Facilitated diffusion differs from simple diffusion through an open channel in the following important way: although the rate of diffusion through an open channel increases proportionately with the concentration of the diffusing substance, in facilitated diffusion, the rate of diffusion approaches a maximum, called the Vmax, as the concentration of the substance increases (figure on the right). The main reason that facilitated diffusion reaches a maximal transport capacity, the Vmax, is due to the time it takes for the transporting protein to go through conformational changes to open to the opposite side of the membrane and return back to its original conformation (figure on the left).
example: Glucose
Simple Diffusion
osmosis
example
osmotic pressure
rates
Fick's Law
membrane permeability
properties
can take place in an open system or across a partition separating 2 systems
inversely related to molecular size
related to temperature
Rapid over short distances, much slower under longer ones.
mvmt continues until molecules are equal everywhere.
molecules move from high to low concentration.
passive process
Physical Isolation
Types
Organelle Membranes
Plasma Membrane
Each cell is surrounded by a plasma membrane that separates it from the extracellular milieu.
Components
The plasma membrane contains enzymes, receptors, and antigens that play central roles in the interaction of the cell with other cells and with hormones and other regulatory agents in the extracellular fluid.
Membrane Proteins
According to some estimates, membrane proteins may make up nearly ⅓ of all proteins coded in human DNA. Each cell has between 10 and 50 different types of proteins inserted into its membranes. Anatomically, membrane proteins are classified into three categories:
Functional Classification
Membrane Enzymes
• enzymes attached to the intracellular surface play an important role in transferring signals from the extracellular compartment to the cytoplasm
- for example, cells lining the lumen of the small intestine are responsible for secreting enzymes involved with the digestion of peptides and carbohydrates
catalyze chemical reactions that take place either on the cell’s external surface or just inside the cell
Structural Proteins
attach cells to the extracellular matrix by linking cytoskeleton fibers to extracellular collagen
and other protein fibers
create cell junctions that hold tissue together, such as tight junctions and gap junctions
• connect the membrane to the cytoskeleton to maintain the shape of the cell
Membrane-Spanning Proteins
Function in protein-mediated transport
Membrane Receptors
are part of the body’s chemical signaling system
• the binding of a receptor with its ligand usually triggers another event at the membrane
- such as activation of an enzyme
- can play an important role in some forms of vesicle transport
Membrane Transporters
move molecules across the membranes and the
transporter proteins can be subdivided into two categories:
Channel Proteins
water-filled passageways that directly link the intracellular and extracellular compartments
An ion’s charge makes it difficult for the ion to move across the lipid bilayer. However, the rapid movement of ions across a membrane is an important aspect of many cellular functions. This movement occurs through selective ion channels. Ion channels are integral proteins spanning the width of the plasma membrane and are normally composed of several polypeptide subunits. Certain specific stimuli cause the protein subunits to open a “gate”, creating an aqueous channel through which the ions can move rapidly from one side of the membrane to the other. Most ion channels usually open in response to a specific stimulus.
Ion channels can be classified according to their gating mechanisms or by the signals that make them open or close. There are voltage-gated channels, ligand-gated channels, and some channels are always open and are referred to as nongated “leak” channels.
Ion channels are often selective for various anions and cations but the relationship between the amino acid composition of the channel protein and ion selectivity of the channel is still under intense investigation.
Open/"Leak"
nongated ion channels are always open and are responsible for the influx of Na+
and the efflux of K+ when the excitable cell is in its resting state (figure A)
• ion movement is determined by an electro-chemical gradient
• these are referred to as “leak” channels
Gated
Mechanically (not for ions?)
Ligand/Chemical
. ligand-gated ion channels are directly activated by the binding of a specific agonist,
a chemical substance, to a membrane receptor (figure B)
• when the agonist (ligand) binds to its receptor, the associated ion channels can
either open or close to permit or block the movement of specific ions across the
cell membrane
- the ligand can be a neurotransmitter arriving from the extracellular medium
- it may be an intracellular second messenger, produced in response to some cell
activity or hormone action, that reaches the ion channel from inside of the cell
Voltage
. voltage-gated ion channels are sensitive to the voltage difference across the cell membrane (figure C)
• these channels are typically closed and they open when the membrane potential changes beyond a certain threshold value
Carrier Proteins
bind to the substrates that they carry but never form a direct connection between the ICF and ECF
Change conformation
Antiport
Symport
Uniport
Structural Classification
Found in
-cell junctions
-cytosketons
Peripheral Proteins
. peripheral proteins do not span the entire width of the cell membrane
• they attach themselves loosely to transmembrane proteins or to polar heads of the
phospholipids
• peripheral proteins include enzymes and some structural binding proteins that
anchor the cytoskeleton to the membrane
Lipid Anchored Proteins
. lipid-anchored proteins are covalently bound to lipid tails that insert themselves into the bilayer
• many are found in association with membrane sphingolipids, leading to the
formation of specialized patches of the membrane called lipid rafts
Integral Proteins
extend all the way across the
cell membrane
• they are tightly bound into the membrane because the 20-25 amino acids in the
α-helix segments that pass through the bilayer are nonpolar
• these amino acids create noncovalent interactions with the lipid tails of the
phospholipids in the membrane
• integral proteins are classified according to how many transmembrane segments
they have
• the amino acid chains protrude into the cytoplasm and the extracellular fluid
Intracellular Loops
phosphate groups can attach to the intracellular loops.
Extracellular Loops
Membrane-spanning integral proteins have loops of peptide chains that extend into the cytoplasm and extracellular fluid. Carbohydrates can attach to the extracellular loops,
Carbohydrates
Glycoproteins
Lipids
Sphngolipids
the major membrane lipid
another significant component of the membrane which have a
fatty acid tail, but their heads may be either phospholipids or glycolipids
Phospho-lipid or GLycolipid Head
Fatty Acid Tail
Lipid Rafts
Aggregates of sphingolipids and cholesterol can form stable microdomains that diffuse laterally in the phospholipid bilayer and form lipid rafts. I
t appears the lipid rafts may mediate the temporary assembly of complexes of intracellular signaling proteins, as well as the sorting of plasma membrane proteins for internalization.
In the above image, sphingolipids (orange) are longer than phospholipids and stick up above the phospholipids of the membrane (black). A lipid-anchored enzyme, placental alkaline phosphatase (yellow), is almost always associated with a lipid raft.
Phostophlipids
Products
Glycolipids
Lipid Bilayer
. In an aqueous environment, phospholipids tend to form structures that allow the fatty acyl chains to be kept away from contact with water. One such structure is the lipid bilayer. An electron micrograph of a lipid membrane is shown on the right (PM = plasma membrane, M = mitochondria, ER = endoplasmic reticulum).
Fluid Mosaic Model
glycolipids and glycoproteins are formed when carbohydrates bind to proteins and lipids on the extracellular surface
protein molecules of various kinds are inserted into and through the phospholipid bilayer
the phospholipids are arranged in a bilayer → creating a semi-permeable membrane
the hydrophobic lipid tails are “hidden” in the center of the membrane
the hydrophilic phosphate heads face the aqueous solutions inside and outside the cell
Two Fatty Acid Tails
- the fatty acid tail is nonpolar and hydrophobic
another significant component of the membrane which have a
fatty acid tail, but their heads may be either phospholipids or glycolipids
Glycerol-Phosphate Head
- the glycerol-phosphate head of the molecule is polar and hydrophilic
Cholesterol
, which are mostly hydrophobic, insert themselves between the hydrophilic heads of phospholipids
• cholesterol helps make the membranes impermeable to small water-soluble molecules and keeps membranes flexible over a wide range of temperatures
Functions
The plasma membrane serves as a permeability barrier that allows the cell to maintain a cytoplasmic composition far different from the composition of the extracellular fluid.
Distribution of Solutes between compartments
changes when water is added
tonicity
hypertonic
hypotonic
isotoníc
osmolality
osmolarity
Total Body Water
The relationships between volumes of the various body fluid compartments are shown for a normal young healthy male individual weighing 70 kg (2.2 lbs = 1 kg).
Intracellular Fluid
0.4 x BW = 28 L
Extracellular Fluid
0.2 x BW = 14 L
The watery internal environment of multicellular animals is called the extracellular fluid (also known as the interstitial fluid) which surrounds all the cells of the body.
ECF includes the water contained within bones and dense connective tissue, as well as cerebrospinal fluid (CSF)
FUNCTION: Extracellular fluid (ECF) serves as the transition between an organism’s external environment and the intracellular fluid inside cells.
Because extracellular fluid is a buffer zone between the outside world and most cells of the body, elaborate physiological processes have evolved to keep its composition relatively stable.
When the ECF composition varies outside its normal range of values, compensatory mechanisms activate and return the fluid to the normal state.
If the body fails to maintain homeostasis, then normal function is disrupted and a disease state, or pathological condition may result.
Plasma
blood plasma is the remaining 25% of the ECF volume (about 3.5 L)
• the composition the interstitial fluid is almost in equilibrium with plasma except for a higher plasma protein concentration
albumen is the predominant plasma protein (plasma osmotic pressure = 28 mm Hg, interstitial osmotic pressure = 8 mm Hg)
Interstitial Fluid
Interstitial fluid urrounds the cells in the various tissues of the body and accounts for 75% of the ECF volume
• this averages between about 10 L to 14 L (.75 of 0.2xBW)
♥ under some pathological conditions, additional fluid may accumulate in the interstitial space (referred to as a “third space”) and results in “pitting” edema