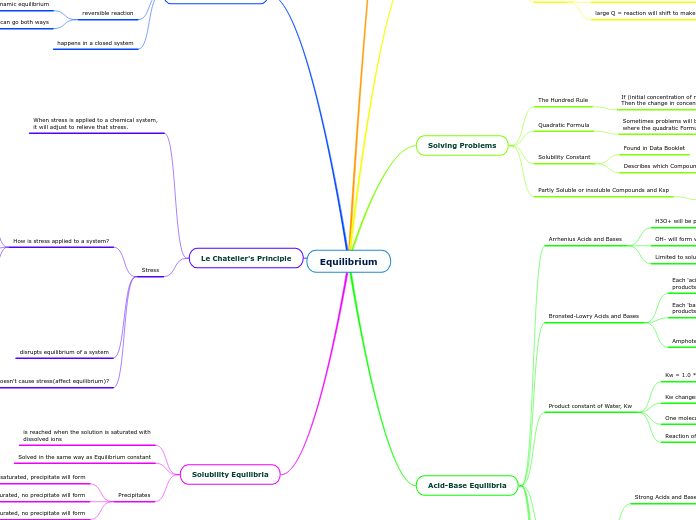
Equilibrium
Precipitates
Q = Ksp , solution is saturated, no precipitate will form
Q < Ksp, solution is unsaturated, no precipitate will form
Q > Ksp, solution is supersaturated, precipitate will form
Solved in the same way as Equilibrium constant
is reached when the solution is saturated with
dissolved ions
Le Chatelier's Principle
Stress
what doesn't cause stress(affect equilibrium)?
Addition of an Inert Gas
Addition of Catalyst
disrupts equilibrium of a system
How is stress applied to a system?
Pressure/Volume
applies to gaseous equilibrium systems
Decrease in air pressure (increase in volume) will
cause the reaction to proceed in the direction which
has the greatest moles of gas
Increase in air pressure (decrease in volume) will cause reaction to proceed in the direction which has the fewest
moles of gas
Concentration
Removal of a reactant or product causes the reaction
to react in a way that produces the removed substance
Addition of reactant or product causes the reaction
to react in a way that consumes the added substance
Temperature
The equilibrium constant stays constant only if
temperature is constant
When heat is removed from system, the
exothermic reaction is favoured
When heat is added to system, the endothermic
reaction is favoured
When stress is applied to a chemical system,
it will adjust to relieve that stress.
Dynamic Equilibrium
happens in a closed system
reversible reaction
reactions equation can go both ways
occurs at equal rates in dynamic equilibrium
Types of Equilibria
Solubility Equilibria
Phase Equilibria
Chemical Reaction Equilibria
Acid-Base Equilibria
Ka, Kb, and Kw
Ka * Kb = Kw
A stronger base (large Kb) will have a weaker conj. acid(small Ka)
A stronger acid(large Ka) will have a weaker conj. base(small Kb)
This equation can be rearranged to solve for what you need
Base Ionization Constant
Weak Bases have a small Ka
Stronger Bases have a large Ka
Acid Ionization Constant
Weak acids have small Ka
Stronger acids have large Ka
Acid Base Strength
Weak Acids and Bases
A weak base will react to create a strong conjugate acid
A weak acid will react to create a strong conjugate base
Do not ionize completely
Strong Acids and Bases
Strong bases will have weak conjugate acids
Strong acids will have waek conjugate bases
Mostly ion sin solution
No Equilibrium
Ionize almost 100% in Water
Product constant of Water, Kw
Reaction of water and water
One molecule acts as an acid, the other as a base
Kw changes with temperature
Kw can be calculated by finding the product of the concentration of Hydronium ions by Hydroxide ions
Kw = 1.0 *10^-14 @ 25 Celcius
Bronsted-Lowry Acids and Bases
Amphoteric
Water is amphoteric
acts as both an acid and a base
Each 'base' on the reactants side has a conjugate acid on the products side
A 'base' takes a Hydrogen atom in the reaction, conjugate acid is formed on the products side.
Each 'acid' on the reactants side has a conjugate base on the products side
An 'acid' gives away its Hydrogen atom in the reaction, conjugate base is formed on the products side
Arrhenius Acids and Bases
Limited to solutions where water is the solvent
OH- will form when a base reacts with water
H3O+ will be produced when and acid reacts in water
Solving Problems
Partly Soluble or insoluble Compounds and Ksp
We write their Ksp using the product of the concentrations of their ions.
Solubility Constant
Describes which Compounds are soluble or not
Found in Data Booklet
Quadratic Formula
Sometimes problems will be set up in a way
where the quadratic Formula must be used.
The Hundred Rule
If (initial concentration of reactant)/(Keq) > 100
Then the change in concentration can be discarded
Reaction Quotient
Q values
large Q = reaction will shift to make reactants
small Q = reaction will shift to make products
Q = 1 , system is at equilibrium
can be used to figure out if a system is at equilibria
measures amounts of reactants and products at a given point
during the reaction
calculated in the same way as the Equilibrium Constant
Equilibrium Constant(Keq)
determines amounts of products and reactants at equilibrium
dimensionless
Keq = (products concentration)/(reactants concentration)
Has different subscripts depending on situation, may need to be solved differently.
Homogeneous reactions
all reactants and products are the in the same phase
Heterogeneous reactions
Reactants and products are not all the same phase
ICE table
used to solve equilibrium problems
Keq value
large Keq = Products favored
small Keq = reactants favoured
K = 1 , neither products or reactants favoured